NCERT Solutions for Class 8 Science Curiosity provide students with a clear, step-by-step approach to understanding complex scientific concepts in an easy and engaging manner. Designed according to the latest CBSE curriculum, these solutions cover all chapters in detail, ensuring that learners not only find accurate answers to textbook questions but also develop strong problem-solving skills. Whether it’s Physics, Chemistry, or Biology, the explanations are presented in a way that nurtures curiosity and encourages deeper learning. This guide is a valuable resource for exam preparation, homework help, and building a solid foundation in science.
Presenting the comprehensive table of contents for NCERT Solutions for Class 8 Science Curiosity, designed to enhance your understanding and spark your curiosity in the subject!
Table of Contents

We are pleased to offer in-depth NCERT Solutions for Class 8 Science Curiosity. Let’s delve in and build a strong understanding of scientific concepts together!
Chapter 1 – Exploring the Investigative World of Science
Exercise Questions and Answers
Q1: Why is one side of a puri thinner than the other?
A1: One side of a puri may be thinner due to uneven rolling of the dough or the way it is dropped into hot oil. When fried, the dough puffs up as water inside turns to steam, pushing the dough outward. If one side is thinner, it expands more, creating a thinner layer compared to the thicker side. Experimenting with dough thickness or dropping methods (e.g., vertical vs. angled) can test this (Page 6).
Q2: Are there more grains of sand on all the beaches and deserts of the world, or more stars in our galaxy?
A2: Estimating this involves comparing large quantities. A single beach may contain billions of sand grains (e.g., a cubic meter of sand has ~1 billion grains). Earth’s beaches and deserts could have ~10^22 to 10^23 grains. The Milky Way has ~100-400 billion stars (~10^11 to 4×10^11). Sand grains likely outnumber stars due to the vast volume of Earth’s beaches and deserts, but precise calculations require more data (Page 1).
Q3: Why has nature created such a vast variety of plants and animals?
A3: The vast variety of plants and animals results from evolution through sexual reproduction, where genetic mixing creates diverse traits. This diversity allows species to adapt to different environments (e.g., deserts, forests, oceans), enhancing survival. Ecosystems benefit from this variety, as each species plays a role in maintaining balance (Pages 1, 5).
Q4: Is there such a question that makes you curious about the world? Write it here!
A4: Example question: Why do some clouds produce rain while others don’t? This sparks curiosity about weather patterns, water cycles, and atmospheric conditions, encouraging observation and investigation (Page 1).
Q5 (Implied from Discover, design, and debate): Design an experiment to investigate why a puri puffs up and why one side is thinner.
A5:
- Question: What factors affect a puri’s puffing and why is one side thinner?
- Hypothesis: Thinner dough or specific dropping methods cause uneven puffing.
- Experiment:
- Prepare dough circles of different thicknesses (e.g., 1 mm, 2 mm, 3 mm) using the same flour (maida).
- Heat oil to a constant temperature (e.g., 180°C).
- Drop dough in two ways: vertically and at an angle.
- Observe and measure: Does it puff up (yes/no)? Time to puff (seconds)? Thickness of each side (mm).
- Control: Use the same oil, temperature, and flour type, changing only one variable (thickness or dropping method) at a time.
- Expected Results: Thinner dough may puff faster but have a thinner side due to greater steam expansion. Vertical dropping may create more uniform puffing than angled dropping (Page 6).
Chapter 2 – The Invisible Living World: Beyond Our Naked Eye
Exercise Questions and Answers (Keep the Curiosity Alive)
Q1: Various parts of a cell are given below. Write them in the appropriate places in the following diagram.
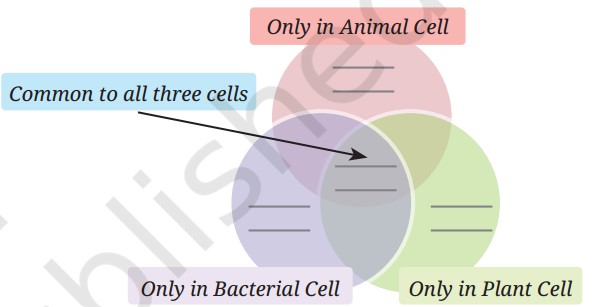
Common to all three cells: Nucleus, Cytoplasm, Cell membrane
Only in Plant Cell: Cell wall, Chloroplast
Only in Bacterial Cell: Nucleoid
A1:
- Animal Cell: Cell membrane, Cytoplasm, Nucleus
- Plant Cell: Cell membrane, Cytoplasm, Nucleus, Cell wall, Chloroplast
- Bacterial Cell: Cell membrane, Cytoplasm, Nucleoid, Cell wall
Q2: Anandi took two test tubes and marked them A and B. She put two spoonfuls of sugar solution in each of the test tubes. In test tube B, she added a spoonful of yeast. Then she attached two incompletely inflated balloons to the mouth of each test tube. She kept the set-up in a warm place, away from sunlight.
(i) What do you predict will happen after 3-4 days? She observed that the balloon attached to test tube B was inflated. What can be a possible explanation for this?
(a) Water evaporated in test tube B and filled the balloon with water vapour.
(b) The warm atmosphere expanded the air inside test tube B, which inflated the balloon.
(c) Yeast produced a gas inside test tube B which inflated the balloon.
(d) Sugar reacted with warm air, which produced gas, eventually inflating the balloon.

A2: Prediction: After 3-4 days, the balloon on test tube B will inflate, while test tube A’s balloon will not.
Observation and Explanation: The balloon on test tube B inflated because yeast, a fungus, ferments sugar, producing carbon dioxide gas that fills the balloon. Test tube A, without yeast, produces no gas, so its balloon remains uninflated (Page 13).
Answer: (c) Yeast produced a gas inside test tube B which inflated the balloon. Options (a), (b), and (d) are incorrect because:
(a) Water evaporation produces minimal vapor, insufficient to inflate a balloon significantly.
(b) Warmth alone cannot expand air enough to inflate the balloon noticeably.
(d) Sugar does not react with warm air to produce gas; fermentation requires a microorganism like yeast.
This is supported by the description of yeast fermentation , where yeast breaks down sugar in warm conditions, releasing carbon dioxide.
(ii) She took another test tube, 1/4 filled with lime water. She removed the balloon from test tube B in such a manner that the gas inside the balloon did not escape. She attached the balloon to the test tube with lime water and shook it well. What do you think she wants to find out?
A2 (ii): Anandi wants to confirm that the gas produced by yeast is carbon dioxide. When carbon dioxide is passed into lime water (calcium hydroxide), it reacts to form calcium carbonate, turning the lime water milky. This test identifies the gas as carbon dioxide (Page 14).
Q3: A farmer was planting wheat crops in his field. He added nitrogen-rich fertiliser to the soil to get a good yield of crops. In the neighbouring field, another farmer was growing bean crops, but she preferred not to add nitrogen fertiliser to get healthy crops. Can you think of the reasons?
A3: The farmer growing beans did not add nitrogen fertilizer because bean crops (legumes) have root nodules containing Rhizobium bacteria. These bacteria fix nitrogen from the air into compounds that enrich the soil, promoting healthy plant growth without additional fertilizers (Page 15).
Q4: Shehald dug two pits, A and B, in her garden. In pit A, she put fruit and vegetable peels and mixed it with dried leaves. In pit B, she dumped the same kind of waste without mixing it with dried leaves. She covered both pits with soil and observed after 3 weeks. What is she trying to test?
A4: Shehald is testing the effect of mixing dried leaves with fruit and vegetable peels on decomposition. Pit A, with mixed leaves, likely decomposes faster due to increased carbon content, aiding microbial activity (fungi, bacteria) to form nutrient-rich manure. Pit B, without leaves, decomposes slower due to less favorable conditions for microbes (Page 12).
Q5: Identify the following microorganisms:
(i) I live in every kind of environment and inside your gut.
(ii) I make bread and cakes soft and fluffy.
(iii) I live in the roots of pulse crops and provide nutrients for their growth.
A5:
- (i) Bacteria (e.g., gut bacteria like Lactobacillus), as they live in diverse environments, including the human gut, aiding digestion (Pages 11, 14).
- (ii) Yeast, a fungus that ferments sugar, releasing carbon dioxide to make bread and cakes soft and fluffy (Pages 13-14).
- (iii) Rhizobium, a bacterium living in root nodules of legumes, fixing nitrogen to provide nutrients for plant growth (Page 15).
Q6: Devise an experiment to test that microorganisms need optimal temperature, air, and moisture for their growth.
A6:
- Question: Do microorganisms need optimal temperature, air, and moisture to grow?
- Hypothesis: Microorganisms grow best in warm, moist conditions with air access.
- Experiment:
- Take four glass containers (A, B, C, D).
- In each, place 50 g of moist soil with vegetable peels.
- Container A: Keep at 25-30°C (warm), uncovered (air access), with moist soil.
- Container B: Keep at 5°C (cold, e.g., refrigerator), uncovered, with moist soil.
- Container C: Keep at 25-30°C, sealed (no air), with moist soil.
- Container D: Keep at 25-30°C, uncovered, with dry soil.
- After 2 weeks, observe decomposition (e.g., formation of dark manure) and use a microscope to check for microbial growth (e.g., fungi, bacteria).
- Prediction: Container A will show the most decomposition and microbial growth due to optimal warmth, air, and moisture. Containers B, C, and D will show less growth due to suboptimal conditions (cold, no air, or dry) (Pages 11-12).
Q7: Take 2 slices of bread. Place one slice in a plate near the sink. Place the other slice in the refrigerator. Compare after three days. Note your observations. Give reasons for your observations.
A7:
- Observations: After three days, the bread near the sink likely shows powdery or cotton-like microbial growth (e.g., mold) due to warmth, moisture, and air exposure. The bread in the refrigerator shows little or no growth, remaining mostly unchanged.
- Reasons: Microorganisms like fungi thrive in warm, moist conditions near the sink, promoting mold growth. The cold refrigerator (5°C) slows microbial activity, inhibiting growth (Pages 11, 14).
Q8: A student observed that when curd is left out for a day, it becomes more sour. What can be two possible explanations for this observation?
A8:
- Bacterial Activity: Lactobacillus bacteria in curd continue to ferment lactose (milk sugar) into lactic acid when left at room temperature, increasing sourness (Page 15).
- Temperature Effect: Warm conditions enhance bacterial growth and fermentation, producing more lactic acid, making the curd sourer compared to refrigerated curd, where cold slows bacterial activity (Page 14).
Q9: Observe the set-up given in Fig. 2.15 and answer the following questions.
(i) What happens to the sugar solution in flask A?
(ii) What do you observe in test tube B after four hours? Why do you think this happened?
(iii) What would happen if yeast was not added in flask A?
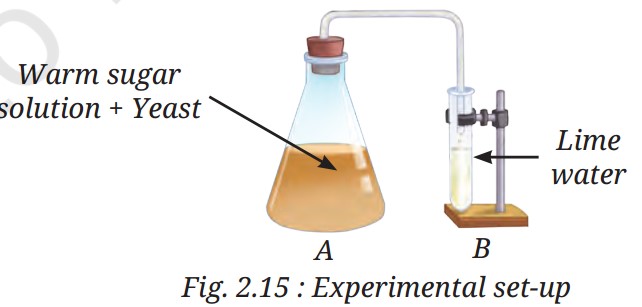
A9:
(iii) If yeast was not added to flask A, no fermentation would occur, so no carbon dioxide would be produced. Test tube B’s lime water would remain clear, as no gas would enter to react (Page 14).
(i) In flask A, yeast ferments the sugar solution, producing carbon dioxide gas and alcohol, causing bubbles and a change in smell (Pages 13-14).
(ii) After four hours, test tube B (containing lime water) turns milky because carbon dioxide from flask A reacts with lime water (calcium hydroxide) to form calcium carbonate, a milky precipitate (Page 14).
Chapter 3 Health: The Ultimate Treasure
Exercise Questions and Answers
Q1: Group the diseases shown in the images as communicable or non-communicable.
Cold and flu, Typhoid, Diabetes, Asthma, Chickenpox

A1:
- Communicable diseases: Cold and flu, Typhoid, Chickenpox
- Non-communicable diseases: Diabetes, Asthma
Q2: From the options given below, identify the non-communicable diseases.
(i) Typhoid, (ii) Asthma, (iii) Diabetes, (iv) Measles
(a) (i) and (ii), (b) (ii) and (iii), (c) (i) and (iv), (d) (ii) and (iv)
A2: (b) (ii) and (iii)
Asthma and Diabetes are non-communicable diseases, while Typhoid and Measles are communicable.
Q3: There is a flu outbreak in your school. Several classmates are absent, while some are still coming to school coughing and sneezing.
(i) What immediate actions should the school take to prevent further spread?
(ii) If your classmate who shares the bench with you starts showing symptoms of the flu, how can you respond in a considerate way without being rude or hurtful?
(iii) How can you protect yourself and others from getting infected in this situation?
A3:
(i) The school should:
- Encourage sick students to stay home and recover.
- Promote handwashing and use of sanitizers.
- Enforce mask-wearing in crowded areas.
- Sanitize classrooms and common areas regularly.
- Educate students on covering their mouth/nose while coughing or sneezing.
(ii) Respond considerately by:
- Kindly suggesting they rest at home to recover quickly, e.g., “I hope you feel better soon! Maybe resting at home could help.”
- Offering to share class notes to support their recovery.
- Maintaining distance politely without making them feel isolated.
(iii) To protect yourself and others:
- Wear a mask and maintain physical distance.
- Wash hands frequently with soap and water.
- Avoid touching your face.
- Use tissues or elbow to cover coughs/sneezes.
- Avoid sharing personal items like water bottles or pens.
Q4: Your family is planning to travel to another city where malaria is prevalent.
(i) What precautions should you take before, during, and after the trip?
(ii) How can you explain the importance of mosquito nets or repellents to your sibling?
(iii) What could happen if travellers ignore health advisories in such areas?
A4:
(i) Precautions:
- Before: Consult a doctor for anti-malarial medication; get vaccinated if available; pack mosquito repellents and nets.
- During: Use mosquito nets while sleeping; apply repellents; wear long-sleeved clothing; avoid stagnant water areas.
- After: Monitor for symptoms like fever or fatigue; seek medical help if symptoms appear.
(ii) Explain to sibling:
- “Mosquito nets and repellents keep mosquitoes away, which carry malaria, a serious disease that can make you very sick. Using them is like wearing a helmet for safety—it protects us from harm.”
(iii) Consequences of ignoring advisories:
- Increased risk of contracting malaria, leading to fever, fatigue, or severe complications.
- Potential spread of the disease to others upon return.
- Higher medical costs and prolonged recovery time.
Q5: Your uncle has started smoking just to fit in with his friends, even though it is well known that smoking can seriously harm health and even cause death.
(i) What would you say to him to make him stop, without being rude?
(ii) What would you do if your friend offers you a cigarette at a party?
(iii) How can schools help prevent students from indulging in such harmful habits?
A5:
(i) To uncle:
- “I really care about you, and I’ve read that smoking can cause serious health problems like cancer. I’d love to see you stay healthy for a long time. Maybe we can find other ways to hang out with your friends?”
(ii) If offered a cigarette:
- Politely decline: “No thanks, I’m good. I’d rather stick to healthy habits.”
- Change the topic or suggest another activity to avoid pressure.
(iii) Schools can help by:
- Conducting awareness campaigns on the dangers of smoking.
- Organizing activities like sports or clubs to engage students positively.
- Inviting health professionals to discuss the impact of harmful substances.
- Enforcing strict no-smoking policies on campus.
Q6: Saniya claims to her friend Vinita that “Antibiotics can cure any infection, so we don’t need to worry about diseases.” What question(s) can Vinita ask her to help Saniya understand that her statement is incorrect?
A6: Vinita could ask:
- “Did you know antibiotics only work against bacterial infections, not viruses like the flu or COVID-19?”
- “What about antibiotic resistance? If we misuse antibiotics, don’t they stop working over time?”
- “How do you think vaccines or lifestyle changes help prevent diseases, since antibiotics can’t cure everything?”
Q7: The following table contains information about the number of dengue cases reported in a hospital over a period of one year:
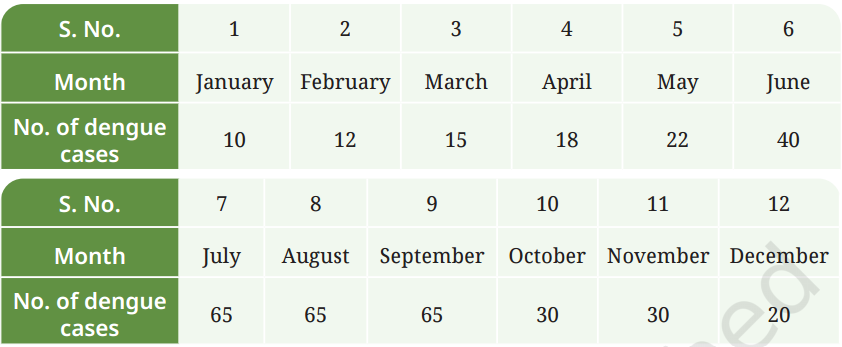
Make a bar graph of the number of cases on the Y-axis and the month on the X-axis. Critically analyse your findings and answer the following:
(i) In which three months were the dengue cases highest?
(ii) In which month(s) were the cases lowest?
(iii) What natural or environmental factors during the peak months might contribute to the increase in dengue cases?
(iv) Suggest a few preventive steps that the community or government can take before the peak season to reduce the spread of dengue.
A7:
Bar Graph:
Below is a textual representation of the bar graph :
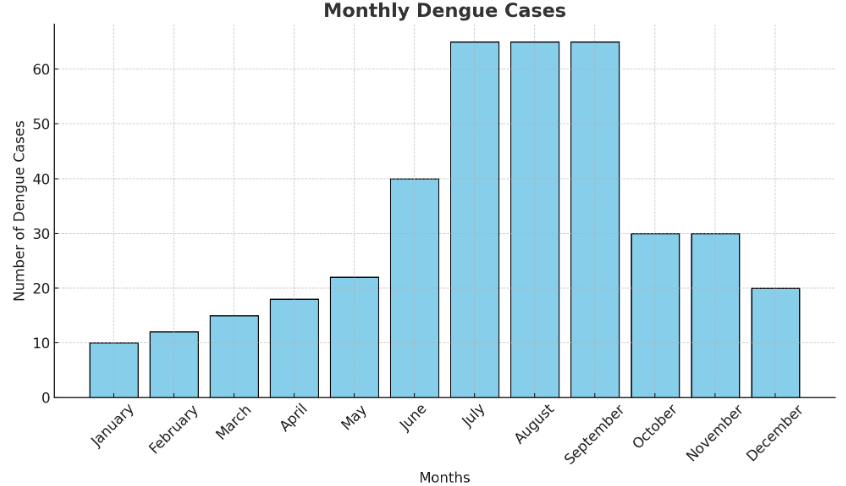
- X-axis: Months (January to December)
- Y-axis: Number of dengue cases
- Data: January (10), February (12), March (15), April (18), May (22), June (40), July (65), August (65), September (65), October (30), November (30), December (20)
(i) Highest three months: July, August, September (65 cases each).
(ii) Lowest month(s): January (10 cases).
(iii) Environmental factors:
- Monsoon season (July–September) brings heavy rainfall, leading to stagnant water, ideal for mosquito breeding.
- Warm temperatures during these months favor mosquito activity and reproduction.
- Poor drainage systems in communities increase breeding sites.
(iv) Preventive steps: - Conduct awareness campaigns on dengue prevention before monsoon.
- Clear stagnant water from drains, pots, and open containers.
- Use mosquito larvicides in water bodies and fogging in high-risk areas.
- Distribute mosquito nets and repellents to residents.
Q8: Imagine you are in charge of a school health campaign. What key messages would you use to reduce communicable and non-communicable diseases?
A8: Key messages:
- For Communicable Diseases: “Wash hands regularly, cover coughs and sneezes, and get vaccinated to stop the spread of infections like flu and typhoid.”
- For Non-Communicable Diseases: “Eat a balanced diet, exercise daily, manage stress, and avoid smoking to prevent diabetes, obesity, and heart disease.”
- General: “Keep your surroundings clean and prioritize mental health by spending time with loved ones.”
Q9: It is recommended that we should not take an antibiotic for a viral infection like a cold, a cough, or flu. Can you provide the possible reason for this recommendation?
A9: Antibiotics target bacteria, not viruses. Using antibiotics for viral infections is ineffective and can contribute to antibiotic resistance, making bacterial infections harder to treat in the future.
Q10: Which disease(s) among the following may spread if drinking water gets contaminated by the excreta from an infected person?
Hepatitis A, Tuberculosis, Poliomyelitis, Cholera, Chickenpox
A10: Diseases that may spread through contaminated drinking water:
- Hepatitis A
- Cholera
- Poliomyelitis
Tuberculosis and Chickenpox do not spread through contaminated water.
Q11: When our body encounters a pathogen for the first time, the immune response is generally low but on exposure to the same pathogen again, the immune response is much more compared to the first exposure. Why is it so?
A11: The first exposure to a pathogen triggers the immune system to produce antibodies and memory cells, which is a slower process. On subsequent exposures, memory cells recognize the pathogen quickly, leading to a faster and stronger immune response.
Chapter 4 – Electricity: Magnetic and Heating Effects
Exercise Questions and Answers
Q1: Fill in the blanks:
(i) The solution used in a Voltaic cell is called ________.
(ii) A current carrying coil behaves like a ________.
A1:
(i) Electrolyte
(ii) Magnet (or Electromagnet)
Q2: Choose the correct option:
(i) Dry cells are less portable compared to Voltaic cells. (True/False)
(ii) A coil becomes an electromagnet only when electric current flows through it. (True/False)
(iii) An electromagnet, using a single cell, attracts more iron paper clips than the same electromagnet with a battery of 2 cells. (True/False)
A2:
(i) False (Dry cells are more portable than Voltaic cells.)
(ii) True
(iii) False (A battery of 2 cells provides more current, increasing the electromagnet’s strength and attracting more clips.)
Q3: An electric current flows through a nichrome wire for a short time.
(i) The wire becomes warm.
(ii) A magnetic compass placed below the wire is deflected.
Choose the correct option:
(a) Only (i) is correct
(b) Only (ii) is correct
(c) Both (i) and (ii) are correct
(d) Both (i) and (ii) are not correct
A3: (c) Both (i) and (ii) are correct
The wire becomes warm due to the heating effect of electric current, and the compass deflects due to the magnetic effect of the current.
Q4: Match the items in Column A with those in Column B.
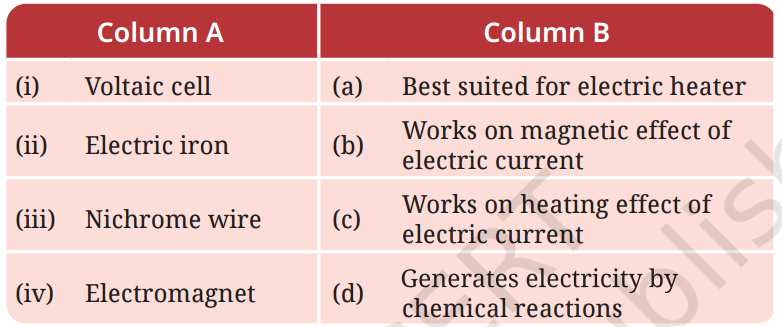
A4: Here’s the correct matching:
(i) Voltaic cell → (d) chemical reactions
(ii) Electric iron → (c) Works on heating effect of electric current
(iii) Nichrome wire → (a) Best suited for electric heater
(iv) Electromagnet → (b) Works on magnetic effect of electric current
Q5: Nichrome wire is commonly used in electrical heating
devices because it
(i) is a good conductor of electricity.
(ii) generates more heat for a given current.
(iii) is cheaper than copper.
(iv) is an insulator of electricity.
A5: The correct answer is: (ii) generates more heat for a given current.
Nichrome has high electrical resistance and can withstand high temperatures without oxidizing, which makes it ideal for heating elements.
Q6: Electric heating devices (like an electric heater or a stove) are often considered more convenient than traditional heating methods (like burning firewood or charcoal). Give reason(s) to support this statement considering societal impact.
A6: Electric heating devices are considered more convenient than traditional heating methods because:
- Cleaner for the environment – They do not produce smoke, ash, or harmful gases, reducing air pollution and health hazards.
- Time-saving – They heat quickly and require no time for gathering fuel or starting a fire.
- Easy to operate – Just switch on the device; no need for constant monitoring or adding fuel.
- Safety – Lower risk of burns from open flames and no sparks that could cause accidental fires.
- Space-friendly – Can be used indoors without the need for chimneys or ventilation for smoke.
- Reduced deforestation – No need to cut down trees for firewood, helping conserve forests.
Q7: Look at the Fig. 4.4a.

If the compass placed near the coil deflects:
(i) Draw an arrow on the diagram to show the path of the electric current.
(ii) Explain why the compass needle moves when current flows.
(iii) Predict what would happen to the deflection if you reverse the battery terminals.
A7: Here’s the solution for each part:
(i) Arrow showing current direction:
In the given diagram, the battery’s positive terminal is at the right and the negative terminal is at the left.
So, the electric current flows from the positive terminal (+) → through the wire to end B → through the coil towards end A → back to the negative terminal (–).
You would draw an arrow along the wire showing this path.
(ii) Why the compass needle moves:
When current flows through the coil, it produces a magnetic field around it.
The compass needle is a small magnet, so the coil’s magnetic field exerts a force on it, causing it to deflect from its usual north–south direction.
(iii) If the battery terminals are reversed:
Reversing the battery changes the direction of current in the coil.
This also reverses the coil’s magnetic polarity, so the compass needle will deflect in the opposite direction compared to before.
Q8: Suppose Sumana forgets to move the switch of her lifting electromagnet model to OFF position. After some time, the iron nail no longer picks up the iron paper clips, but the wire wrapped around the iron nail is still warm. Why did the lifting electromagnet stop lifting the clips? Give possible reasons.
A8:
Possible reasons:
- Cell/Battery Depletion: The cell or battery may have run out of charge, stopping the current flow and thus the magnetic field, causing the electromagnet to lose its ability to attract clips.
- Overheating: The wire remains warm due to the heating effect of the current before the cell depleted, as resistance in the wire converts electrical energy to heat.
- Circuit Issue: A loose connection or wire damage could have interrupted the current, stopping the magnetic effect while residual heat remains in the wire.
Q9: In Fig. 4.11, in which case the LED will glow when the switch is closed?
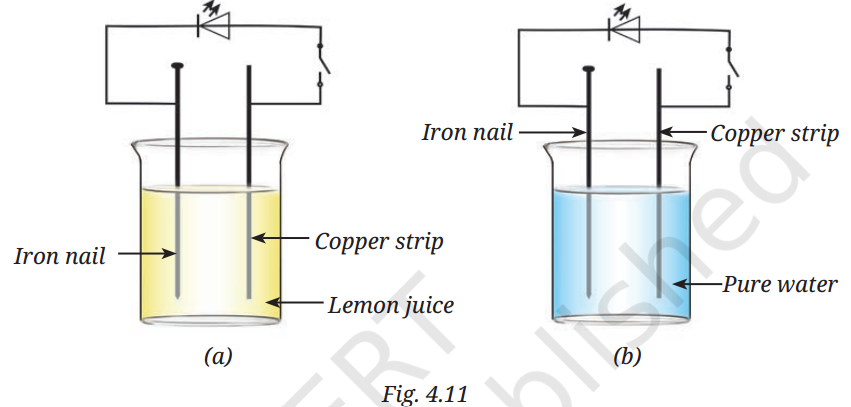
A9: The LED will glow in case (a) — when the electrodes are dipped in lemon juice.
Reason: In case (b), pure water is a poor conductor because it has very few ions, so the circuit will not complete, and the LED will not glow.
Lemon juice contains acids (citric acid), which produce ions in the solution, making it a good conductor of electricity.
Q10: Neha keeps the coil exactly the same as in activity 4.4 but slides the iron nail out, leaving only the coiled wire. Will the coil still deflect the compass? If yes, will the deflection be more or less than before?
A10: Yes, the coil will still deflect the compass even without the iron nail inside.
Reason: When current flows through the coiled wire, it produces a magnetic field, and this magnetic field can influence the compass needle.
Deflection: The deflection will be less than before because the iron nail acted as a soft iron core, which increased the strength of the magnetic field (by concentrating the magnetic lines of force). Without it, the magnetic field is weaker, so the compass needle will not deflect as much.
Q11: We have four coils, of similar shape and size, made up from iron, copper, aluminium, and nichrome as shown in Fig. 4.12.
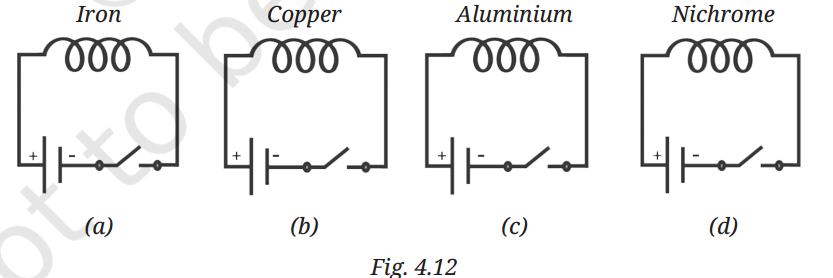
When current is passed through the coils, compass needles placed near the coils will show deflection.
(i) Only in circuit (a)
(ii) Only in circuits (a) and (b)
(iii) Only in circuits (a), (b), and (c)
(iv) In all four circuits.
A11: The correct answer is: (iv) In all four circuits.
Reason:
Any wire carrying an electric current produces a magnetic field, regardless of the material (iron, copper, aluminium, or nichrome). This magnetic field will cause a nearby compass needle to deflect. The difference between materials only affects the strength of the field — ferromagnetic materials like iron make the field stronger when used as a core, but the effect is present for all conducting coils.
Chapter 5 – Exploring Forces
Exercise Questions and Answers
Q1: Match items in Column A with the items in Column B.
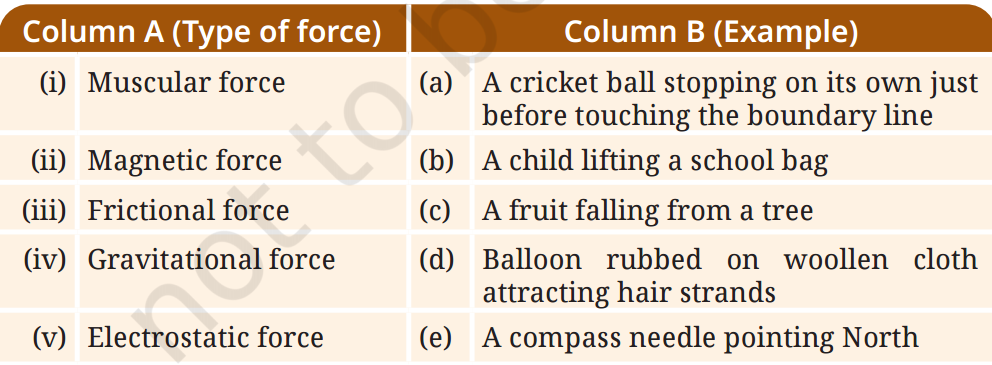
A1: Here’s the correct matching:
(i) Muscular force → (b) A child lifting a school bag
(ii) Magnetic force → (e) A compass needle pointing North
(iii) Frictional force → (a) A cricket ball stopping on its own just before touching the boundary line
(iv) Gravitational force → (c) A fruit falling from a tree
(v) Electrostatic force → (d) Balloon rubbed on woollen cloth attracting hair strands
Q2: State whether the following statements are True or False.
(i) A force is always required to change the speed of motion of an object.
(ii) Due to friction, the speed of the ball rolling on a flat ground increases.
(iii) There is no force between two charged objects placed at a small distance apart.
A2:
(i) True (A force is needed to change speed, as per the effects of force.)
(ii) False (Friction opposes motion, reducing the speed of a rolling ball.)
(iii) False (Charged objects exert an electrostatic force, attracting or repelling each other.)
Q3: Two balloons rubbed with a woollen cloth are brought near each other. What would happen and why?
A3: The balloons repel each other because they acquire similar charges (e.g., both positive or both negative) when rubbed with the woollen cloth. Like charges repel, as demonstrated in Activity 5.7.
Q4: When you drop a coin in a glass of water, it sinks, but when you place a bigger wooden block in water, it floats. Explain.
A4: The coin sinks because its weight (gravitational force) is greater than the buoyant force exerted by the water. The wooden block floats because its weight is equal to or less than the buoyant force, as the wood is less dense and displaces enough water to balance its weight (Archimedes’ Principle).
Q5: If a ball is thrown upwards, it slows down, stops momentarily, and then falls back to the ground. Name the forces acting on the ball and specify their directions.
(i) During its upward motion
(ii) During its downward motion
(iii) At its topmost position.
A5:
(i) Upward motion: Gravitational force (downward) opposes the ball’s motion, slowing it down. Air friction (downward) also opposes the motion.
(ii) Downward motion: Gravitational force (downward) accelerates the ball toward the Earth. Air friction (upward) slightly opposes the motion.
(iii) Topmost position: Gravitational force (downward) acts on the ball, causing it to begin falling. Air friction is negligible as the ball is momentarily stationary.
Q6: A ball is released from point P and moves along an inclined plane, then along a horizontal surface as shown in Fig. 5.16. It comes to a stop at point A on the horizontal surface. Think of a way so that when the ball is released from the same point P it stops (i) before the point A (ii) after crossing the point A.
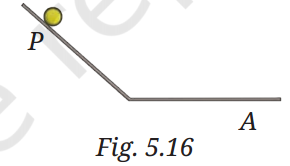
A6:
(i) Stop before point A: Increase friction on the horizontal surface by placing a rough material (e.g., sandpaper or cloth) before point A. This increases the frictional force, stopping the ball sooner.
(ii) Stop after crossing point A: Reduce friction by making the horizontal surface smoother (e.g., using a polished or lubricated surface). This decreases the frictional force, allowing the ball to travel farther past point A.
Q7: Why do we sometimes slip on smooth surfaces like ice or polished floors? Explain.
A7: Smooth surfaces like ice or polished floors have fewer surface irregularities, reducing frictional force. This low friction makes it harder to maintain grip, causing slipping when walking or applying force.
Q8: Is any force being applied to an object in non-uniform motion?
A8: Yes, an object in non-uniform motion (changing speed or direction) experiences a net force. This force, such as gravity, friction, or an applied push/pull, causes the acceleration or deceleration.
Q9: The weight of an object on the Moon becomes one-sixth of its weight on the Earth. What causes this change? Does the mass of the object also become one-sixth of its mass on the Earth?
A9: The weight on the Moon is one-sixth that on Earth because the Moon’s gravitational force is one-sixth that of Earth’s. Weight is the gravitational force (W = m × g), and since g is smaller on the Moon, weight decreases. The mass of the object remains the same, as mass is the amount of matter and does not depend on location.
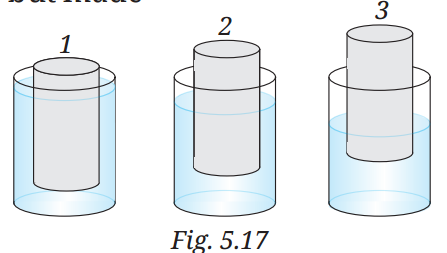
Q10: Three objects 1, 2, and 3 of the same size and shape, but made of different materials are placed in water. They dip to different depths as shown in Fig. 5.17. If the weights of the three objects are w₁, w₂, and w₃, respectively, then
(i) w₁ = w₂ = w₃
(ii) w₁ > w₂ > w₃
(iii) w₁ < w₂ < w₃
(iv) w₁ > w₃ > w₂
A10: (ii) w₁ > w₂ > w₃
Objects dipping to different depths indicate different buoyancies due to their densities. The object that sinks deepest (highest density) has the greatest weight, as it displaces less water and experiences less buoyant force. Assuming object 1 sinks deepest, object 2 is partially submerged, and object 3 floats highest, their weights follow w₁ > w₂ > w₃.
Chapter 6 – Pressure, Winds, Storms, and Cyclones
Exercise Questions and Answers
Q1:
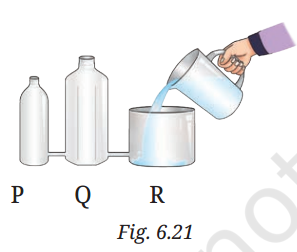
(i) Look at Fig. 6.21 carefully. Vessel R is filled with water.
When pouring of water is stopped, the level of water will
be ________.
(a) the highest in vessel P
(b) the highest in vessel Q
(c) the highest in vessel R
(d) equal in all three vessels
(ii) A rubber sucker (M) is pressed on a flat smooth surface and an identical sucker (N) is pressed on a rough surface:
(a) Both M and N will stick to their surfaces.
(b) Both M and N will not stick to their surfaces.
(c) M will stick but N will not stick.
(d) M will not stick but N will stick
(iii) A water tank is placed on the roof of a building at a height ‘H’. To get water with more pressure on the ground floor, one has to
(a) increase the height ‘H’ at which the tank is placed.
(b) decrease the height ‘H’ at which the tank is placed.
(c) replace the tank with another tank of the same height that can hold more water.
(d) replace the tank with another tank of the same height that can hold less water.
(iv)
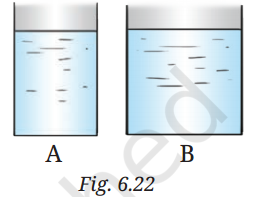
Two vessels, A and B contain water up to the same level as shown in Fig. 6.22. PA and PB is the pressure at the bottom of the vessels. FA and FB is the force exerted by the water at the bottom of the vessels A and B.
(a) PA = PB, FA = FB
(b) PA = PB, FA < FB
(c) PA < PB, FA = FB
(d) PA > PB, FA > FB
A1: The correct statements are:
(i) (d) equal in all three vessels
Reason: The vessels are interconnected, so water will settle at the same level in P, Q, and R due to the principle of communicating vessels.
(ii) (c) M will stick but N will not stick
Reason: A rubber sucker works only on a smooth surface where an airtight seal can form; on a rough surface, air leaks in, preventing suction.
(iii) The correct answer is: (a) increase the height ‘H’ at which the tank is placed
Reason: Water pressure at the outlet depends on the height of the water column above it (hydrostatic pressure). The greater the height HH, the greater the pressure on the ground floor.
(iv) The correct answer is: (b) PA=PB, FA<FB
Reason:
Force (FF) is pressure × area of the bottom surface. Vessel B has a larger base area, so FB>FA
Pressure (PP) at the bottom depends only on the height of the water column, not the shape or width of the container. Since both have the same water level, PA=PB.
Q2: State whether the following statements are True [T] or False [F].
(i) Air flows from a region of higher pressure to a region of lower pressure. [ ]
(ii) Liquids exert pressure only at the bottom of a container. [ ]
(iii) Weather is stormy at the eye of a cyclone. [ ]
(iv) During a thunderstorm, it is safer to be in a car. [ ]
A2: (i) True (T) ✅ — Air naturally moves from high-pressure regions to low-pressure regions.
(ii) False (F) ❌ — Liquids exert pressure in all directions, not just at the bottom.
(iii) False (F) ❌ — The eye of a cyclone is calm; the stormy weather is around the eye wall.
(iv) True (T) ✅ — A car acts as a Faraday cage, protecting you from lightning during a thunderstorm.
Q3:
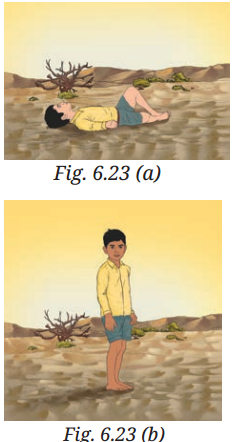
Fig. 6.23 a shows a boy lying horizontally, and Fig. 6.23b shows the boy standing vertically on
loose sand bed. In which case does the boy sink more in sand? Give reasons.
A3: Conclusion:
The boy sinks more in the sand when standing vertically (Fig. 6.23b). This is because the pressure is higher due to the smaller contact area, causing the sand to yield more under his weight compared to when he is lying horizontally, where the pressure is lower due to the larger contact area.
Reason:
The sinking depends on the pressure exerted on the sand. Higher pressure (from standing) compresses the loose sand more, allowing the boy to sink deeper, while lower pressure (from lying down) distributes the weight, reducing the depth of sinking.
Q4: An elephant stands on four feet. If the area covered by one foot is 0.25 m2 calculate the pressure exerted by the elephant on the ground if its weight is 20000N.
A4: Let’s calculate step-by-step:
Given:
- Area of one foot = 0.25 m^2
- Number of feet = 4
- Total area in contact with ground = 4×0.25=1.0 m^2
- Weight (Force) F = 20000N
Formula: Pressure=Force/Area
Substitute values: P=20000/1.0=20000 Pa
Answer: The pressure exerted = 20000 Pa
Q5: There are two boats, A and B. Boat A has a base area of 7 m², and 5 persons are seated in it. Boat B has a base area of 3.5 m², and 3 persons are seated in it. If each person has a weight of 700 N, find out which boat will experience more pressure on its base and by how much?
A5:
- Boat A: Total force = 5 persons × 700 N = 3500 N. Base area = 7 m².
Pressure = Force / Area = 3500 N / 7 m² = 500 N/m². - Boat B: Total force = 3 persons × 700 N = 2100 N. Base area = 3.5 m².
Pressure = Force / Area = 2100 N / 3.5 m² = 600 N/m². - Comparison: Boat B experiences more pressure (600 N/m²) than Boat A (500 N/m²).
- Difference: 600 N/m² – 500 N/m² = 100 N/m².
Boat B experiences 100 N/m² more pressure than Boat A.
Q6: Would lightning occur if air and clouds were good conductors of electricity? Give reasons for your answer.
A6: No, lightning would not occur if air and clouds were good conductors of electricity. Lightning occurs because air is an insulator, allowing a buildup of opposite charges in clouds and between clouds and the ground. When the charge difference becomes large, the air’s insulating property breaks down, causing a sudden flow of charges (lightning). If air and clouds were good conductors, charges would flow continuously, preventing the buildup needed for lightning.
Q7: What will happen to the two identical balloons A and B as shown in Fig. 6.24 when water is filled into the bottle up to a certain height? Will both the balloons bulge? If yes, will they bulge equally? Explain your answer.
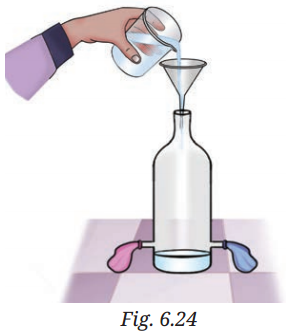
A7: Both balloons will bulge, and they will bulge equally. The pressure exerted by the water depends on the height of the water column, not the volume or diameter of the container. Since the water is filled to the same height in both bottles, the pressure at the bottom is the same, causing both balloons to bulge to the same extent.
Q8: Explain how a storm becomes a cyclone.
A8: A storm becomes a cyclone when it forms over warm ocean waters. Warm, moist air rises, creating a low-pressure area. As the air rises, water vapor condenses, releasing heat that further lowers the pressure. Surrounding air rushes in, begins to spin due to Earth’s rotation (Coriolis effect), and forms a large, rotating system of clouds, winds, and rain. This system, with a calm center (eye) and high-speed winds, is a cyclone.
Q9: Fig. 6.25 shows trees along the sea coast in a summer afternoon. Identify which side is land – A or B. Explain your answer.

A9: Side A is land. In a summer afternoon, the land heats up faster than the sea, creating a low-pressure area over the land. Air moves from the high-pressure sea (side B) to the low-pressure land (side A), causing a sea breeze. The trees lean toward side A due to the wind blowing from the sea (side B) to the land (side A).
Q10: Describe an activity to show that air flows from a region of high pressure to a region of low pressure.
A10:
- Take two balloons and a straw. Inflate one balloon and secure one end of the straw inside it with a rubber band. Attach the other end of the straw to an uninflated balloon.
- Observe that air flows from the inflated balloon (high-pressure region) to the uninflated balloon (low-pressure region), causing the uninflated balloon to inflate and the inflated balloon to shrink until the pressures equalize.
This demonstrates that air moves from a high-pressure region to a low-pressure region.
Q11: What is a thunderstorm? Explain the process of its formation.
A11: A thunderstorm is a storm with lightning and thunder, often accompanied by heavy rain or hail.
Formation: Warm, moist air rises due to heating, creating a low-pressure area. Cooler air flows in, rises, and cools, condensing water vapor into clouds. Strong upward and downward winds cause water droplets and ice particles to rub, generating static electric charges. Positive charges accumulate in the upper cloud, and negative charges in the lower cloud or ground. When the charge difference overcomes air’s insulating property, a sudden charge flow produces lightning, and the rapid air expansion causes thunder.
Q12: Explain the process that causes lightning.
A12: Lightning is caused by a buildup of opposite electric charges in clouds or between clouds and the ground. Warm, moist air rises, cools, and forms clouds. Strong winds cause water droplets and ice particles to rub, generating static charges—positive charges in the upper cloud and negative charges in the lower cloud or ground. Air, normally an insulator, prevents charge flow until the charge difference becomes large, breaking down air’s insulation. This results in a sudden flow of charges, producing a bright flash called lightning.
Q13: Explain why holes are made in banners and hoardings.
A13: Holes are made in banners and hoardings to allow wind to pass through, reducing wind pressure on the surface. Without holes, high-speed winds create a low-pressure area on one side, increasing the pressure difference across the banner, which could tear it or cause it to collapse. Holes minimize this pressure difference, making the banner more stable.
Chapter 7 – Particulate Nature of Matter
Exercise Questions and Answers
Q1: Choose the correct option. The primary difference between solids and liquids is that the constituent particles are:
(i) closely packed in solids, while they are stationary in liquids.
(ii) far apart in solids and have fixed position in liquids.
(iii) always moving in solids and have fixed position in liquids.
(iv) closely packed in solids and move past each other in liquids.
A1: (iv) closely packed in solids and move past each other in liquids.
Explanation: Solids have closely packed particles with strong interparticle forces, restricting movement to vibrations. Liquids have weaker forces, allowing particles to move past each other, giving liquids no fixed shape.
Q2: Which of the following statements are true? Correct the false statements.
(i) Melting ice into water is an example of the transformation of a solid into a liquid.
(ii) Melting process involves a decrease in interparticle attractions during the transformation.
(iii) Solids have a fixed shape and a fixed volume.
(iv) The interparticle interactions in solids are very strong, and the interparticle spaces are very small.
(v) When we heat camphor in one corner of a room, the fragrance reaches all corners of the room.
(vi) On heating, we are adding energy to the camphor, and the energy is released as a smell.
A2:
(i) True: Melting ice into water is a transformation from solid to liquid.
(ii) False: The melting process involves particles gaining enough thermal energy to overcome interparticle attractions, not a decrease in the attractions themselves.
Corrected: The melting process involves particles gaining thermal energy to overcome interparticle attractions, allowing them to move apart.
(iii) True: Solids have a fixed shape and volume due to strong interparticle forces and close packing.
(iv) True: Solids have strong interparticle interactions and minimal interparticle spaces.
(v) True (T) ✅ — Camphor sublimates, and its vapour diffuses through the air, carrying the fragrance to all parts of the room.
(vi) False (F) ❌ — On heating, we add energy to the camphor, causing it to change into vapour; the smell is not energy release, but the vapour molecules reaching our nose.
Q3: Choose the correct answer with justification. If we could remove all the constituent particles from a chair, what would happen?
(i) Nothing will change.
(ii) The chair will weigh less due to lost particles.
(iii) Nothing of the chair will remain.
A3: Correct Answer: (iii) Nothing of the chair will remain.
Justification: A chair, like all objects, is made up of constituent particles such as atoms and molecules, which are held together by various forces (e.g., chemical bonds, intermolecular forces). These particles define the chair’s structure, mass, and physical properties. If we could remove all the constituent particles:
Option (i) Nothing will change: This is incorrect because removing all particles would eliminate the chair’s physical existence, as the chair is fundamentally composed of these particles. Without them, there would be no material or structure left.
Option (ii) The chair will weigh less due to lost particles: This is also incorrect. Weight is a measure of the force exerted by the mass of the chair due to gravity, and mass is determined by the total number of particles. If all particles are removed, the chair would cease to exist, and the concept of weight would no longer apply.
Option (iii) Nothing of the chair will remain: This is correct. Removing all constituent particles would mean disassembling the chair at its most fundamental level, leaving no material or physical form behind. The chair would effectively disappear, as its existence depends entirely on the presence of these particles.
Conclusion– The removal of all constituent particles results in the complete disappearance of the chair, making (iii) the correct answer.
Q4: Why do gases mix easily, while solids do not?
A4: Gases mix easily because the molecules in a gas are far apart and move very quickly in all directions. This high speed and large spacing mean they can spread out and intermingle without much restriction. In solids, the particles are tightly packed in fixed positions and can only vibrate in place, so they cannot move freely to mix with particles of another solid.
Q5: When spilled on the table, milk in a glass tumbler, flows and spreads out, but the glass tumbler stays in the same shape. Justify this statement.
A5: Milk flows and spreads out because it is a liquid, and in liquids, particles are close together but can slide past each other. This allows a liquid to take the shape of its container and flow when poured. The glass tumbler keeps its shape because it is a solid, and in solids, particles are tightly packed in fixed positions, so they can only vibrate but cannot move around. This rigidity makes a solid maintain its own shape.
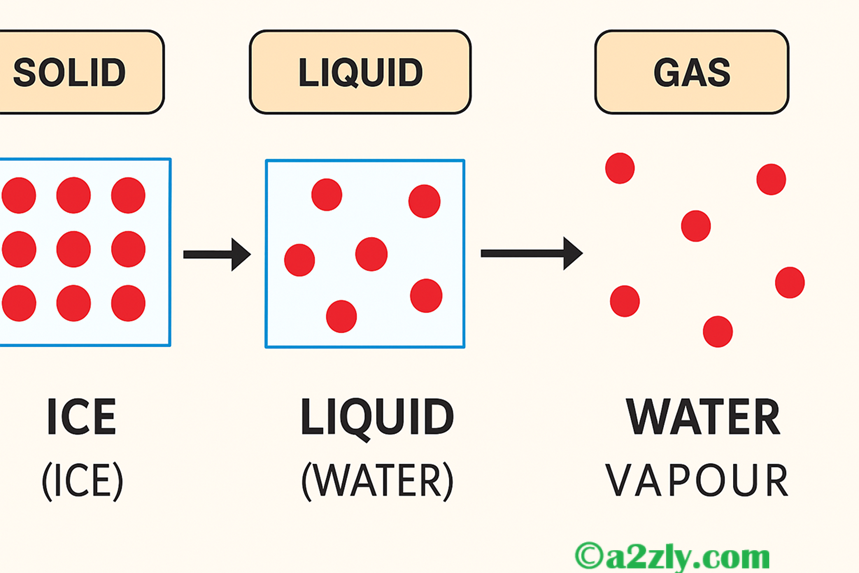
Q6: Represent diagrammatically the changes in the arrangement of particles as ice melts and transforms into water vapour.
A6:
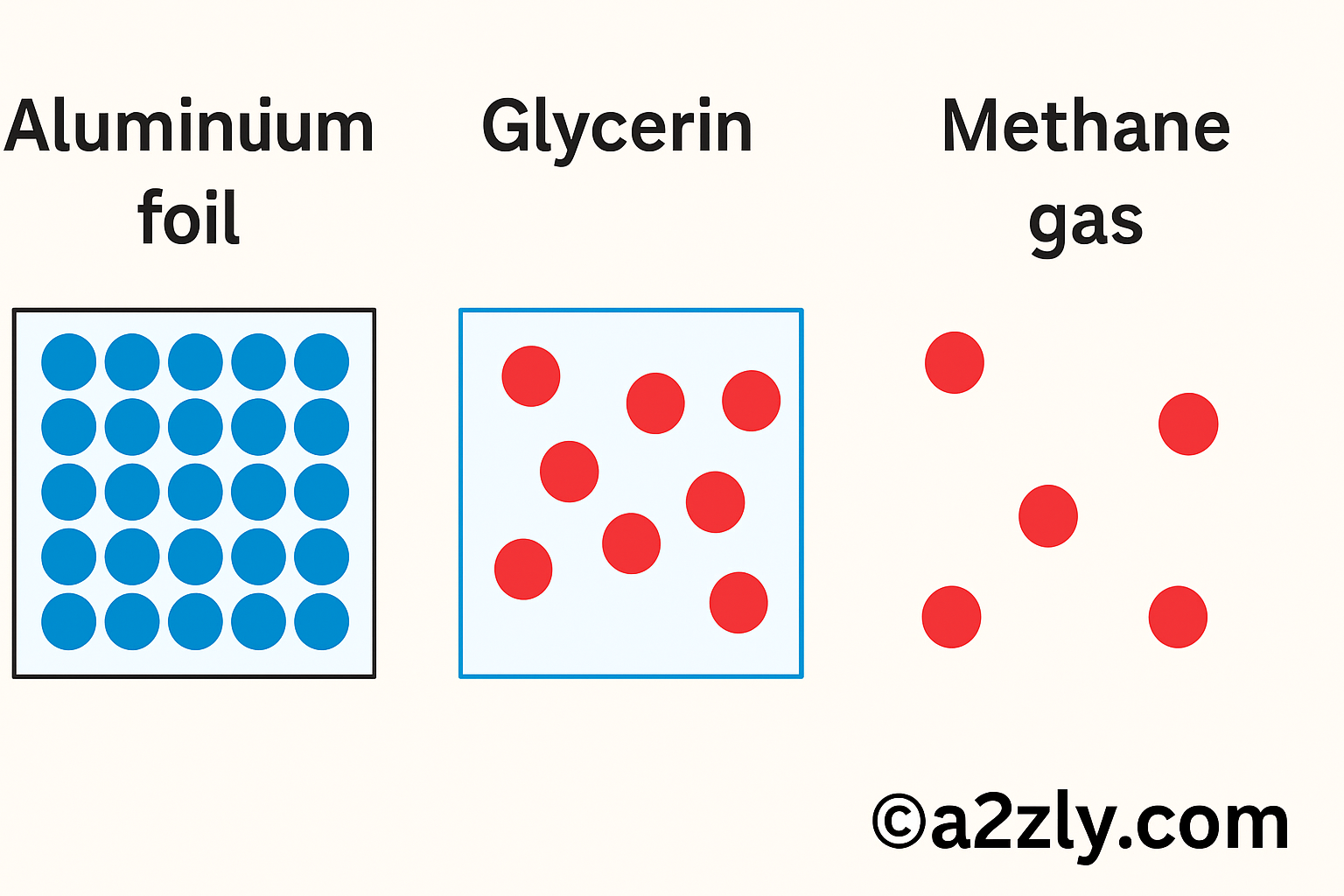
Q7: Draw a picture representing particles present in the following: (i) Aluminium foil (ii) Glycerin (iii) Methane gas
A7:
Q8: Observe Fig. 7.16a, which shows the image of a candle that was just extinguished after burning for some time. Identify the different states of wax in the figure and match them with Fig. 7.16b showing the arrangement of particles.
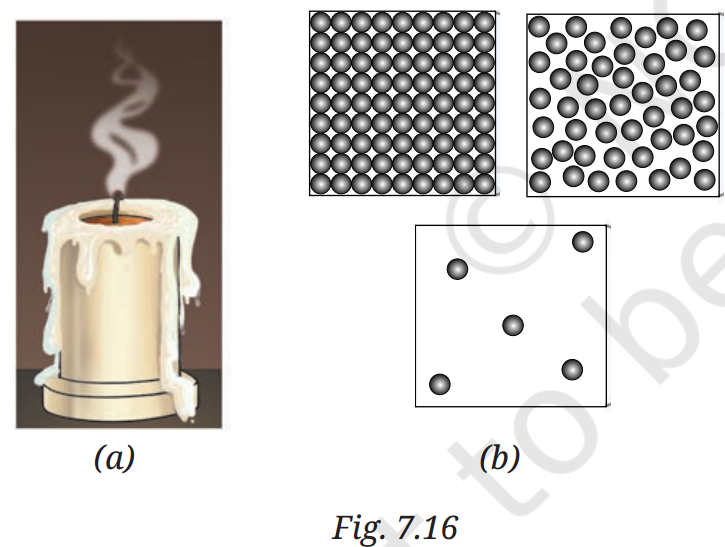
A8: In Fig. 7.16a, the candle shows:
- Solid wax: The unburned wax, with closely packed particles vibrating in fixed positions (matches Fig. 7.16b, solid state).
- Liquid wax: The melted wax around the wick, with particles moving freely within a limited space (matches Fig. 7.16b, liquid state).
- Gaseous wax: The wax vapor that burns, with particles moving freely in all directions (matches Fig. 7.16b, gaseous state).
Q9: Why does the water in the ocean taste salty, even though the salt is not visible?
A9: Ocean water tastes salty because salt dissolves into its constituent particles, which are too small to be seen. These particles spread among water particles, occupying interparticle spaces, and can be detected by taste.
Q10: Grains of rice and rice flour take the shape of the container when placed in different jars. Are they solids or liquids? Explain.
A10: Grains of rice and rice flour are solids. They appear to take the shape of the container because their small particles can flow and settle, but each grain or particle retains its own fixed shape and volume, characteristic of solids, due to strong interparticle forces.
Chapter 8 – Nature of Matter: Elements, Compounds, and Mixtures
Exercise Questions and Answers
Q1: Consider the following reaction where two substances, A and B, combine to form a product C: A + B → C. Assume that A and B cannot be broken down into simpler substances by chemical reactions. Which of the following statements is correct?
(i) A, B, and C are all compounds and only C has a fixed composition.
(ii) C is a compound, and A and B have a fixed composition.
(iii) A and B are compounds, and C has a fixed composition.
(iv) A and B are elements, C is a compound, and has a fixed composition.
A1: (iv) A and B are elements, C is a compound, and has a fixed composition.
Explanation: Since A and B cannot be broken down into simpler substances, they are elements. When they combine to form C, it is a compound with a fixed composition, as compounds are formed by elements chemically combining in a fixed ratio.
Q2: Assertion: Air is a mixture. Reason: A mixture is formed when two or more substances are mixed, without undergoing any chemical change.
(i) Both Assertion and Reason are true and Reason is the correct explanation for Assertion.
(ii) Both Assertion and Reason are true, but Reason is not the correct explanation for Assertion.
(iii) Assertion is true, but Reason is false.
(iv) Assertion is false, but Reason is true.
A2: (i) Both Assertion and Reason are true and Reason is the correct explanation for Assertion.
Explanation: Air is a mixture of gases (nitrogen, oxygen, etc.) that retain their properties and do not undergo chemical changes when mixed. The reason correctly explains why air is a mixture.
Q3: Water, a compound, has different properties compared to those of the elements oxygen and hydrogen from which it is formed. Justify this statement.
A3: Water (H₂O) is a compound formed by the chemical combination of hydrogen and oxygen in a fixed ratio (2:1). Hydrogen is a flammable gas, and oxygen supports combustion, but water is a liquid that extinguishes fire, showing distinct properties. This is because the chemical bonding in water creates a new substance with different characteristics than its constituent elements.
Q4: In which of the following cases are all the examples correctly matched? Give reasons in support of your answer:
(i) Elements – water, nitrogen, iron, air.
(ii) Uniform mixtures – minerals, seawater, bronze, air.
(iii) Pure substances – carbon dioxide, iron, oxygen, sugar.
(iv) Non-uniform mixtures – air, sand, brass, muddy water.
A4: (iii) Pure substances – carbon dioxide, iron, oxygen, sugar.
Reasons:
- (i) Incorrect: Water is a compound, and air is a mixture, not elements. Only nitrogen and iron are elements.
- (ii) Incorrect: Minerals are mostly compounds or elements, not mixtures. Seawater, bronze, and air are uniform mixtures.
- (iii) Correct: Carbon dioxide and sugar are compounds, and iron and oxygen are elements, all of which are pure substances (single type of particle).
- (iv) Incorrect: Air and brass are uniform mixtures, not non-uniform. Sand and muddy water are non-uniform mixtures.
Q5: Iron reacts with moist air to form iron oxide, and magnesium burns in oxygen to form magnesium oxide. Classify all the substances involved in the above reactions as elements, compounds, or mixtures, with justification.
A5:
- Iron: Element – It is a pure substance made of identical iron atoms, not breakable into simpler substances.
- Moist air: Mixture – It is a uniform mixture of gases (nitrogen, oxygen, etc.) and water vapor, with components retaining their properties.
- Iron oxide: Compound – Formed by iron and oxygen chemically combining in a fixed ratio, with different properties (e.g., rust is not magnetic).
- Magnesium: Element – A pure substance made of identical magnesium atoms.
- Oxygen: Element – A pure substance made of identical oxygen atoms or molecules (O₂).
- Magnesium oxide: Compound – Formed by magnesium and oxygen chemically combining, with different properties than its elements (e.g., a white solid, unlike the metal or gas).
Q6: Classify the following as elements, compounds, or mixtures in Table 8.3: Carbon dioxide, sand, seawater, magnesium oxide, muddy water, aluminium, gold, oxygen, rust, iron sulfide, glucose, air, water, fruit juice, nitrogen, sodium chloride, sulfur, hydrogen, baking soda.
A6:
Table 8.3
| Elements | Compounds | Mixtures |
|---|---|---|
| Aluminium | Carbon dioxide | Sand |
| Gold | Magnesium oxide | Seawater |
| Oxygen | Rust (iron oxide) | Muddy water |
| Nitrogen | Iron sulfide | Air |
| Sulfur | Glucose | Fruit juice |
| Hydrogen | Water | |
| Sodium chloride | ||
| Baking soda (sodium bicarbonate) |
Pure substances: Carbon dioxide, magnesium oxide, rust, iron sulfide, glucose, water, sodium chloride, baking soda, aluminium, gold, oxygen, nitrogen, sulfur, hydrogen.
Explanation: Elements are pure substances with one type of atom. Compounds are pure substances with two or more elements chemically combined. Mixtures contain multiple substances retaining their properties. Sand and muddy water are non-uniform mixtures; seawater, air, and fruit juice are uniform mixtures.
Q7: What new substance is formed when a mixture of iron filings and sulfur powder is heated, and how is it different from the original mixture? Also, write the word equation for the reaction.
A7: When a mixture of iron filings and sulfur powder is heated, iron sulfide is formed.
Differences:
- The original mixture (Sample A) has visible black (iron) and yellow (sulfur) particles, is magnetic (iron attracted), and iron reacts with hydrochloric acid to produce hydrogen gas, while sulfur does not.
- Iron sulfide (Sample B) is a black, uniform compound with no magnetic properties, and reacts with hydrochloric acid to produce hydrogen sulfide gas (rotten egg odor).
Word Equation: Iron + Sulfur → Iron sulfide
Q8: Is it possible for a substance to be classified as both an element and a compound? Explain why or why not.
A8: No, a substance cannot be classified as both an element and a compound. An element is a pure substance made of identical atoms that cannot be broken down further (e.g., oxygen, iron). A compound is a pure substance formed by two or more elements chemically combined in a fixed ratio (e.g., water, iron sulfide). By definition, a substance is either one type of atom (element) or a combination of different atoms (compound), making it impossible to be both.
Q9: How would our daily lives be changed if water were not a compound but a mixture of hydrogen and oxygen?
A9: If water were not a compound (H₂O) but simply a mixture of hydrogen and oxygen gases, life would be drastically different — and mostly impossible. Here’s why:
- No liquid water – A mixture of hydrogen and oxygen at room temperature would remain as separate gases, not a liquid, so rivers, lakes, and oceans wouldn’t exist.
- No universal solvent – Water’s unique ability to dissolve many substances (which supports chemical reactions in living organisms) comes from its molecular structure. Without it, processes like digestion, nutrient transport in plants, and cleaning would fail.
- Dangerous to store – Hydrogen and oxygen gases together are highly explosive. A simple spark could ignite them, causing violent explosions. This means even a glass of “water” would be a potential bomb.
- No support for life – All living things rely on water’s chemical properties for metabolism. A mixture of gases couldn’t sustain the biochemical reactions that keep cells alive.
- Weather and climate collapse – Rain, snow, clouds, and the water cycle depend on H₂O’s ability to change states between solid, liquid, and gas. With only gases, there’d be no precipitation and no stable climate patterns.
In short: without water as a compound, Earth would be a dry, lifeless, and dangerous planet.
Q10:
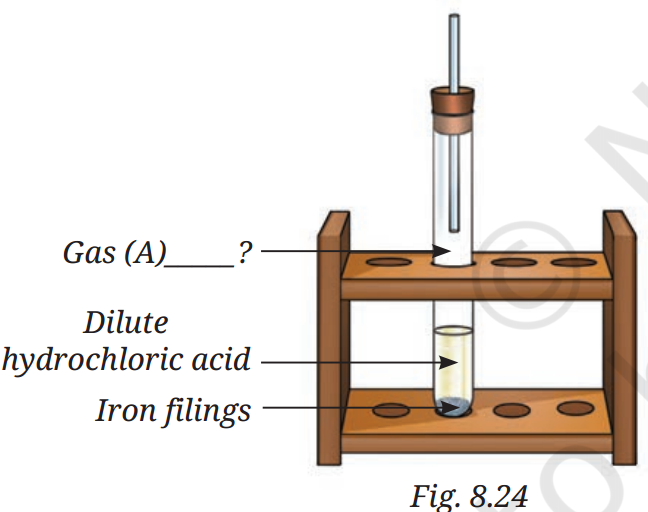
Analyse Fig. 8.24. Identify Gas A. Also, write the word equation of the chemical reaction.
A10: In Fig. 8.24, when dilute hydrochloric acid reacts with iron filings, Gas A is Hydrogen gas (H₂).
Word equation of the reaction:
Iron + Hydrochloric acid ⟶ Iron chloride + Hydrogen
In symbols:
Fe + 2HCl ⟶ FeCl₂ + H₂ ↑
The hydrogen gas is collected in the test tube above.
Q11: Write the names of any two compounds made only from non-metals, and also mention two uses of each of them.
A11: Here are two examples:
Carbon dioxide (CO₂)
Use 1: Used in fire extinguishers. Use 2: Used in carbonated drinks.
Sulphur dioxide (SO₂)
Use 1: Used as a preservative in dried fruits. Use 2: Used in the manufacture of sulphuric acid.
Q12: How can gold be classified as both a mineral and a metal?
A12: Gold can be classified as both a mineral and a metal because:
- As a mineral:
Gold occurs naturally in the Earth’s crust, often found in rocks, veins, and alluvial deposits. When it is present in its natural, pure form, it is considered a mineral because it meets the definition — naturally occurring, inorganic, and having a definite chemical composition (elemental gold, Au). - As a metal:
Gold is also a metallic element with properties such as high malleability, ductility, good electrical conductivity, and metallic lustre. These physical and chemical properties classify it as a metal in the periodic table.
In short, gold in nature is a mineral, and gold as an element is a metal.
Chapter 9 – The Amazing World of Solutes, Solvents, and Solutions
Exercise Questions and Answers
Q1: State whether the statements given below are True [T] or False [F]. Correct the false statement(s).
(i) Oxygen gas is more soluble in hot water rather than in cold water.
(ii) A mixture of sand and water is a solution.
(iii) The amount of space occupied by any object is called its mass.
(iv) An unsaturated solution has more solute dissolved than a saturated solution.
(v) The mixture of different gases in the atmosphere is also a solution.
A1: Here’s the answer with corrections for the false statements:
(i) False (F) – Oxygen gas is more soluble in cold water rather than in hot water.
(ii) False (F) – A mixture of sand and water is not a solution, it is a suspension.
(iii) False (F) – The amount of space occupied by any object is called its volume, not mass.
(iv) False (F) – An unsaturated solution has less solute dissolved than a saturated solution.
(v) True (T) – The mixture of different gases in the atmosphere is indeed a solution (a gaseous solution).
Q2: Fill in the blanks.
(i) The volume of a solid can be measured by the method of displacement, where the solid is submerged in water and the rise in water level is measured.
(ii) The maximum amount of solute dissolved in a solvent at a particular temperature is called solubility at that temperature.
(iii) Generally, the density decreases with increase in temperature.
(iv) The solution in which glucose has completely dissolved in water, and no more glucose can dissolve at a given temperature, is called a saturated solution of glucose.
Q3: You pour oil into a glass containing some water. The oil floats on top. What does this tell you?
(i) Oil is denser than water.
(ii) Water is denser than oil.
(iii) Oil and water have the same density.
(iv) Oil dissolves in water.
A3: (ii) Water is denser than oil.
Explanation: Oil floats on water because it is less dense than water (e.g., oil’s density is ~0.91 g/cm³, water’s is 1 g/cm³). Oil does not dissolve in water, and their densities is not the same.
Q4: A stone sculpture weighs 25 g and has a volume of 90 cm³. Calculate its density and predict whether it will float or sink in water.
A4:
Density Calculation:
Density = Mass / Volume = 25 g / 90 cm³ = 0.278 g/cm³.
Prediction: The stone will float in water because its density (0.278 g/cm³) is less than water’s density (1 g/cm³). Objects with lower density than water float.
Q5: Which one of the following is the most appropriate statement, and why are the other statements not appropriate?
(i) A saturated solution can still dissolve more solute at a given temperature.
(ii) An unsaturated solution has dissolved the maximum amount of solute possible at a given temperature.
(iii) No more solute can be dissolved into the saturated solution at that temperature.
(iv) A saturated solution forms only at high temperatures.
A5: (iii) No more solute can be dissolved into the saturated solution at that temperature.
Explanation:
- (iii) Correct: A saturated solution has dissolved the maximum amount of solute possible at a given temperature, so no more can dissolve without changing conditions.
- (i) Incorrect: A saturated solution cannot dissolve more solute at the same temperature.
- (ii) Incorrect: An unsaturated solution can dissolve more solute, not the maximum amount.
- (iv) Incorrect: Saturated solutions can form at any temperature, not only high temperatures.
Q6: You have a bottle with a volume of 2 liters. You pour 50 mL of water into it. How much more water can the bottle hold?
A6:
Calculation:
2 liters = 2000 mL.
Water poured = 50 mL.
Remaining capacity = 2000 mL – 50 mL = 1950 mL.
Answer: The bottle can hold 1950 mL more water.
Q7: An object has a mass of 400 g and a volume of 40 cm³. What is its density?
A7: Density of the object Density=Mass/Volume
Mass = 400 g, Volume = 40 cm³ Density=400/40=10 g/cm3
Answer: 10 g/cm³
Q8: Analyse Fig. 9.25a and 9.25b. Why does the unpeeled orange float, while the peeled one sinks? Explain.
A8:
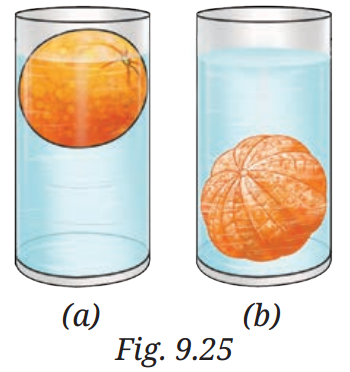
Floating and sinking of oranges (Fig. 9.25a & 9.25b)
Unpeeled orange: The peel contains tiny air pockets, making its average density less than water. This helps it float.
Peeled orange: Removing the peel removes the trapped air, increasing the density so it becomes greater than water’s density, and it sinks.
Reason: Floating or sinking depends on density compared to water — less dense objects float, denser objects sink.
Q9: Object A has a mass of 200 g and a volume of 40 cm³. Object B has a mass of 240 g and a volume of 60 cm³. Which object is denser?
A9: Comparing densities of Object A and B
For Object A: Density=200/40=5 g/cm3
For Object B: Density=240/60=4 g/cm3
Answer: Object A is denser because 5 g/cm3>4 g/cm3
Q10: Reema has a piece of modeling clay that weighs 120 g. She first moulds it into a compact cube that has a volume of 60 cm³. Later, she flattens it into a thin sheet. Predict what will happen to its density.
A10: The density of the modeling clay will remain the same.
Explanation: Density = Mass / Volume. The mass (120 g) remains constant, and changing the shape (from cube to thin sheet) does not affect the volume of the material itself. Thus, the density remains 120 g / 60 cm³ = 2 g/cm³.
Q11: A block of iron has a mass of 600 g and a density of 7.9 g/cm³. What is its volume?
A11:
Calculation:
Density = Mass / Volume.
Volume = Mass / Density = 600 g / 7.9 g/cm³ ≈ 75.95 cm³.
Answer: The volume of the iron block is approximately 75.95 cm³.
Q12: You are provided with an experimental setup as shown in Fig. 9.26a and 9.26b. On keeping the test tube (Fig. 9.26b) in a beaker containing hot water (~70 °C), the water level in the glass tube rises. How does it affect the density?
A12:
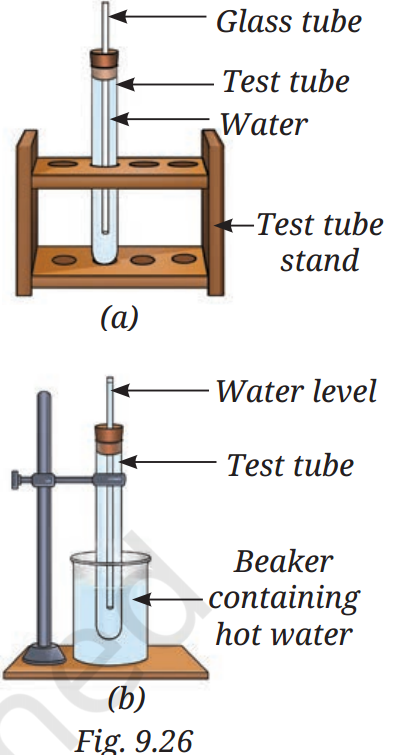
When the test tube in Fig. 9.26(b) is placed in hot water:
- The water inside the test tube absorbs heat.
- On heating, the water molecules move faster and spread farther apart → volume increases (thermal expansion).
- Mass stays the same, but volume increases.
Since Density=Mass/Volume
and volume ↑ while mass stays constant → density decreases.
Final Answer:
The density of the water decreases because heating makes it expand, so the same mass now occupies a larger volume. The rise in water level in the glass tube is due to this expansion.
Chapter 10 – Light: Mirrors and Lenses
Exercise Questions and Answers
Q1: A light ray is incident on a mirror and gets reflected by it (Fig. 10.21). The angle made by the incident ray with the normal to the mirror is 40°. What is the angle made by the reflected ray with the mirror?
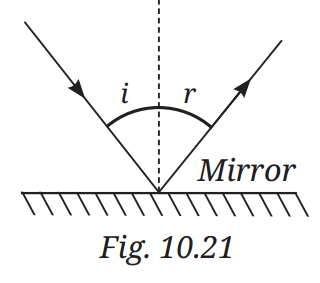
A1: According to the first law of reflection, the angle of incidence (i) equals the angle of reflection (r). If the angle of incidence is 40°, the angle of reflection is also 40°.
Answer: The angle made by the reflected ray with the normal is 40°.
Angle made by the reflected ray with the mirror is 50°.
Q2.
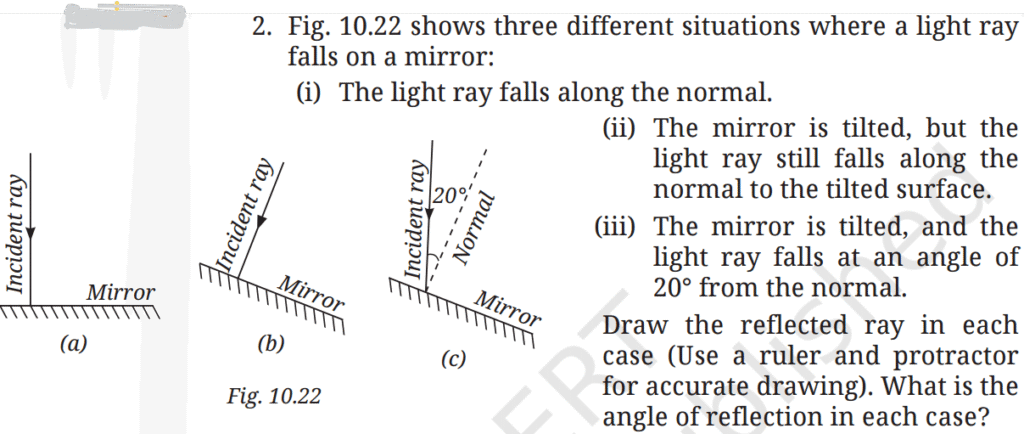
A2: (i) Light ray falls along the normal (Fig. 10.22a)
- Angle of incidence = 0°
- Angle of reflection = 0°
- The ray will be reflected back along the same path.
(ii) Mirror is tilted, but ray still falls along the normal to the tilted surface (Fig. 10.22b)
- Even though the mirror is tilted, the ray is incident along the normal to that tilted mirror.
- Angle of incidence = 0°
- Angle of reflection = 0°
- The ray will be reflected back along the same path (relative to the tilted surface).
(iii) Mirror tilted, light ray falls at 20° from the normal (Fig. 10.22c)
Reflected ray will make 20° with the normal, but in the opposite direction to the incident ray.
Angle of incidence = 20°
Angle of reflection = 20° (on the other side of the normal)

Q3:

A3:Here’s the correct matching for the mirrors shown:
- (i)– Convex mirror → Image appears smaller than the object.
- (ii)– Concave mirror → Image appears larger (magnified) when object is close.
- (iii)- Plane mirror → Image is of the same size as the object.
Q4: In Fig. 10.24, the cap of a sketch pen is placed behind a convex lens, a concave lens, and a flat transparent glass piece – all at the same distance. Match each image with the correct optical element.
A4:

- Image (i): Erect and enlarged → Matches Convex Lens (objects close to a convex lens appear erect and enlarged).
- Image (ii): Erect and diminished → Matches Concave Lens (concave lenses always produce erect, diminished images).
- Image (iii): Same size as the object → Matches Flat Transparent Glass Piece (no bending of light, so the object appears unchanged).
Answer:
(i) Convex Lens
(ii) Concave Lens
(iii) Flat Transparent Glass Piece
Q5: When the light is incident along the normal on the mirror, which of the following statements is true:
(i) Angle of incidence is 90°
(ii) Angle of incidence is 0°
(iii) Angle of reflection is 90°
(iv) No reflection of light takes place in this case
A5:When light is incident along the normal on a mirror, the correct statement is (ii) Angle of incidence is 0°.
Explanation:
“Normal” means perpendicular to the surface. When light hits a mirror along the normal, it has no angle of incidence with the mirror’s surface, resulting in a reflected ray that also travels along the normal.
Q6: Three mirrors — plane, concave and convex are placed in Fig. 10.25. On the basis of the
images of the graph sheet formed in the mirrors, identify the mirrors and write their names above the mirrors.

A6: Let’s carefully observe Fig. 10.25:
- Left mirror – The image of the graph sheet appears smaller than the actual size. This is the property of a convex mirror (diverging, produces diminished image).
- Middle mirror – The image of the graph sheet appears of the same size. This is the property of a plane mirror (produces same-sized image).
- Right mirror – The image of the graph sheet appears magnified. This is the property of a concave mirror (converging, can produce magnified image when object is close).
Q7: In a museum, a woman walks towards a large convex mirror (Fig. 10.26). She will see that:
(i) her erect image keeps decreasing in size.
(ii) her inverted image keeps decreasing in size.
(iii) her inverted image keeps increasing in size and eventually it becomes erect and magnified.
(iv) her erect image keeps increasing in size.
A7:
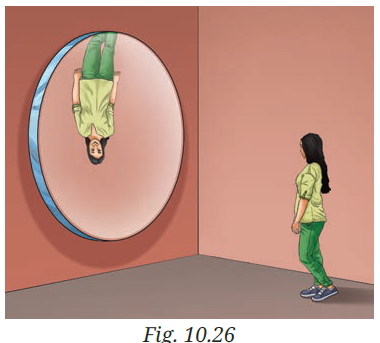
The question is about a large convex mirror.
But in the picture (Fig. 10.26), the artist has drawn the image inverted, which actually happens with a concave mirror, not a convex one.
- Convex mirror → always forms an erect, diminished image (never inverted). As the woman walks closer, the image size increases but always remains erect and smaller than her.
Correct option: (i) her erect image keeps decreasing in size. - Concave mirror (if this was the real situation in the figure) → the image can be inverted or erect depending on distance. If she moves closer, the inverted image enlarges, then eventually becomes erect and magnified when she is within the focal length.
In that case, correct option would be (iii).
So, based on the text of the question (convex mirror) → Answer is (i).
Based on the figure shown (which looks like a concave mirror image) → Answer is (iii).
Q8: Hold a magnifying glass over text and identify the distance where you can see the text bigger than they are written. Now move it away from the text. What do you notice? Which type of lens is a magnifying glass?
A8: Observation with a magnifying glass
- When held close to the text (within focal length):
- The letters look bigger than their actual size.
- The image is erect, virtual, and magnified.
- When you move the glass farther away (beyond focal length):
- The letters no longer appear magnified.
- The image appears inverted and smaller (not useful for reading).
Conclusion– A magnifying glass is a convex lens.
It works as a magnifier only when the object is placed within its focal length.
Q9:

A9: Answer:
(i) Concave mirror → (a)
(ii) Convex mirror → (b)
(iii) Convex lens → (c)
(iv) Concave lens → (d)
Q10: The following question is based on Assertion/Reason.
Assertion: Convex mirrors are preferred for observing the traffic behind us.
Reason: Convex mirrors provide a significantly larger view area than plane mirrors.
Choose the correct option:
(i) Both Assertion and Reason are correct and Reason is the correct explanation for Assertion.
(ii) Both Assertion and Reason are correct but Reason is not the correct explanation for Assertion.
(iii) Assertion is correct but Reason is incorrect.
(iv) Both Assertion and Reason are incorrect.
A10: (i) Both Assertion and Reason are correct and Reason is the correct explanation for Assertion.
Explanation: Convex mirrors form virtual, erect, and diminished images, so they give a wider field of view than plane mirrors. This larger view area (and reduced blind spots) is why convex mirrors are preferred for observing traffic behind us.
Q11:

A11:
Correct Answer:
(ii) Figure (a) indicates a convex mirror and Figure (b) indicates a concave mirror.
Q12: Place a pencil behind a transparent glass tumbler (Fig. 10.28a). Now fill the tumbler halfway with water (Fig. 10.28b). How does the pencil appear when viewed through the water? Explain why its shape appears changed.

A12:
Appearance: The pencil appears bent at the water’s surface when viewed through the tumbler filled halfway with water.
Explanation: This is due to the refraction of light. When light travels from water (a denser medium) to air (a less dense medium), it bends away from the normal, causing the submerged part of the pencil to appear at a different angle than the part in air. This creates the illusion of a bent pencil at the air-water interface (Fig. 10.28b).
Chapter 11 – Keeping Time with the Skies
Exercise Questions and Answers
Q1: State whether the following statements are True or False.
(i) We can only see that part of the Moon which reflects sunlight towards us.
(ii) The shadow of Earth blocks sunlight from reaching the Moon causing phases.
(iii) Calendars are based on various astronomical cycles which repeat in a predictable manner.
(iv) The Moon can only be seen at night.
A1: Here’s the evaluation of the statements:
(i) We can only see that part of the Moon which reflects sunlight towards us.
✅ True
(ii) The shadow of Earth blocks sunlight from reaching the Moon causing phases.
❌ False (Phases are due to changing positions of the Moon and Earth; Earth’s shadow causes lunar eclipses, not phases.)
(iii) Calendars are based on various astronomical cycles which repeat in a predictable manner.
✅ True
(iv) The Moon can only be seen at night.
❌ False (The Moon is often visible during the day also, depending on its phase and position in the sky
Q2: Amol was born on 6th of May on a full Moon day. Does his birthday fall on the full Moon day every year? Explain your answer.
A2: No, Amol’s birthday will not fall on a full Moon day every year. Here’s why:
Sometimes a full Moon may fall close to 6th May, but in most years it will occur on different dates in May (or even late April or early June).
A year (based on Earth’s revolution around the Sun) is about 365 days, while the lunar cycle (time from one full Moon to the next) is about 29.5 days. These two cycles are not in exact sync. That means the date of a full Moon keeps shifting in the calendar every year.
Q3:

A3: Two things shown incorrectly are:
- Cause of the dark part of the Moon –
The figure shows clouds (or Earth’s shadow) covering the Moon to explain its dark part.
This is wrong.
The dark part of the Moon is actually due to the Moon not receiving sunlight there, not because something covers it. - Stars visible in front of the Moon –
In the figure, stars are drawn in front of the Moon (overlapping).
This is wrong because stars are much farther away, so they can never appear in front of the Moon.
Q4:

A4: (i) Match picture labels with phases of the Moon:
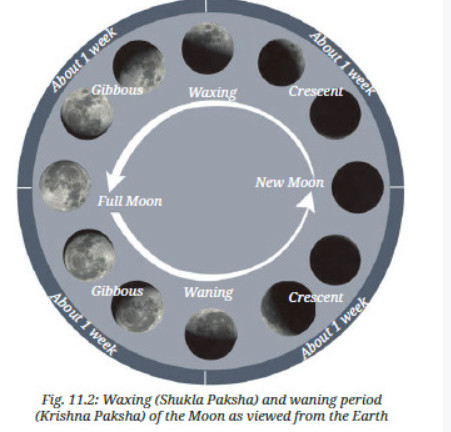
- Three days after New Moon → D (small crescent visible on right side).
- Full Moon → E (complete bright circle).
- Three days after Full Moon → A (slightly dark edge on right, waning gibbous).
- A week after Full Moon → C (half lit on left, last quarter).
- Day of New Moon → B (completely dark).
(ii) Phases of the Moon that are never seen from Earth:
- Picture B – On a new Moon day, only the non-illuminated side of the Moon faces the Earth, so it is not visible from Earth.
- Picture F – This is not a phase of the Moon. So, it can never be seen from Earth
Q5: Malini saw the Moon overhead in the sky at sunset. (i) Draw the phase of the Moon that Malini saw. (ii) Is the Moon in the waxing or the waning phase?
A5: (i) At sunset, the Moon appears overhead only during the first quarter phase, which occurs about one week after the New Moon. In this phase, the right half of the Moon is illuminated while the left half remains dark. Therefore, we should draw a half Moon with the right side bright and the left side shaded.

(ii) It is the waxing phase because it occurs after the New Moon, and the illuminated portion of the Moon is increasing day by day.
Q6: Ravi said, “I saw a crescent Moon, and it was rising in the East, when the Sun was setting.” Kaushalya said, “Once I saw the gibbous Moon during the afternoon in the East.” Who out of the two is telling the truth?
A6: Kaushalya is correct because a gibbous Moon can be seen in the eastern sky during the afternoon.
Ravi’s statement is incorrect because a crescent Moon does not rise in the east at sunset. A crescent Moon appears either just after the New Moon (waxing crescent) or just before the New Moon (waning crescent).
- A waxing crescent is seen after sunset in the western sky, not rising in the east.
- A waning crescent rises before sunrise, not at sunset.
Therefore, Kaushalya’s statement is true, while Ravi’s statement is false.
Q7: Scientific studies show that the Moon is getting farther away from the Earth and slower in its revolution. Will luni-solar calendars need an intercalary month more often or less often?
A7: Luni-solar calendars would need an intercalary month (Adhika Maasa) less often. The reason is simple: an extra month is added because a lunar year (about 354 days) is shorter than a solar year (about 365 days), leaving a gap of roughly 11 days each year.
If the Moon’s revolution slows down, the lunar month—the time between one full cycle of phases—would become longer. A longer lunar month would make the lunar year longer as well.
As a result, the difference between the lunar and solar years would shrink. This mismatch would build up more slowly over time, meaning an extra month would be required less frequently to keep the calendar aligned with the seasons.
Q8: A total of 37 full Moons happen during 3 years in a solar calendar. Show that at least two of the 37 full Moons must happen during the same month of the solar calendar.
A8: Yes, at least two full Moons must occur within the same solar month.
A solar calendar has 12 months per year, so in 3 years there are:
12 × 3 = 36 months.
If there are 37 full Moons in these 36 months, then by necessity, at least one month must contain two full Moons.
Q9: On a particular night, Vaishhali saw the Moon in the sky from sunset to sunrise. What phase of the Moon would she have noticed?
A9: If the Moon is visible from sunset to sunrise, it is up all night, which occurs when it is nearly opposite the Sun in the sky. This corresponds to the full Moon phase, when the Moon rises at sunset and sets at sunrise (Fig. 11.5a, position A).
Answer: Full Moon.
Q10: If we stopped having leap years, in approximately how many years would the Indian Independence Day happen in winter?
A10: A leap year adds about one extra day every four years. Without leap years, the calendar would gradually shift by one day every four years. There are roughly 183 days between 15 August (monsoon season) and mid-February (winter).
So, the time required for this shift would be:
183 × 4 = 732 years . Therefore, in approximately 730–732 years, 15 August would fall during winter if leap years were not included.
Q11: What is the purpose of launching artificial satellites?
A11: Artificial satellites are launched for communication, navigation, weather monitoring, disaster management, and scientific research, such as mapping (e.g., ISRO’s Cartosat) and studying celestial objects (e.g., AstroSat) (Page 16).
Answer: For communication, navigation, weather monitoring, disaster management, and scientific research.
Q12: On which periodic phenomenon are the following measures of time based: (i) day (ii) month (iii) year?
A12:
- (i) Day: Based on Earth’s rotation on its axis, causing the cycle of day and night (mean solar day, ~24 hours) (Activity 11.3).
- (ii) Month: Based on the Moon’s phases, completing a cycle from new Moon to new Moon in ~29.5 days (Page 10).
- (iii) Year: Based on Earth’s revolution around the Sun, taking ~365.25 days, linked to the cycle of seasons (Page 10).
Answer:
(i) Day: Earth’s rotation
(ii) Month: Moon’s phases
(iii) Year: Earth’s revolution around the Sun
Chapter 12 – How Nature Works in Harmony
Exercise Questions and Answers
Q1: Refer to the given diagram (Fig. 12.19) and select the wrong statement.
(i) A community is larger than a population.
(ii) A community is smaller than an ecosystem.
(iii) An ecosystem is part of a community.
A1:
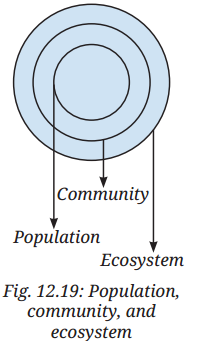
(i) True: A community includes multiple populations of different species in a habitat.
(ii) True: A community is part of an ecosystem, which includes both biotic (community) and abiotic components.
(iii) False: An ecosystem includes a community and abiotic components, not the other way around.
Answer: (iii) An ecosystem is part of a community.
Q2: A population is part of a community. If all decomposers suddenly disappear from a forest ecosystem, what changes do you think would occur? Explain why decomposers are essential.
A2: If decomposers (e.g., fungi, bacteria) disappear from a forest ecosystem:
- Dead plants, animals, and waste would accumulate, as decomposition would stop.
- Nutrients would not be recycled into the soil, reducing soil fertility.
- Plants (producers) would suffer from nutrient deficiency, impacting their growth.
- Herbivores and other consumers dependent on plants would decline due to food scarcity.
- The food web would collapse, disrupting the ecosystem’s balance.
Why decomposers are essential: Decomposers break down dead matter, recycling nutrients (e.g., nitrogen, carbon) into the soil, which plants use for growth. This supports the entire food web and maintains ecosystem stability (Page 11).
Answer: Nutrient recycling would stop, leading to reduced soil fertility, plant decline, and collapse of the food web. Decomposers are essential for breaking down dead matter and recycling nutrients for plant growth.
Q3: Selvam from Cuddalore district, Tamil Nadu, shared that his village was less affected by the 2004 Tsunami compared to nearby villages due to the presence of mangrove forests. This surprised Sarita, Shabnam, and Shijo. They wondered if mangroves were protecting the village. Can you help them understand this?
A3: Mangrove forests in Selvam’s village acted as a natural barrier during the 2004 Tsunami. Their dense roots and trees slowed down strong waves and winds, reducing the tsunami’s impact on the village. Mangroves also stabilize coastlines, prevent erosion, and absorb storm energy, protecting inland areas. This explains why Selvam’s village was less affected compared to nearby villages without mangroves (Page 15, Fig. 12.17).
Answer: Mangroves protected the village by slowing tsunami waves, stabilizing coastlines, and reducing storm impact.
Q4: Look at this food chain: Grass → Grasshopper → Frog → Snake. If frogs disappear from this ecosystem, what will happen to the population of grasshoppers and snakes? Why?
A4:
- Grasshoppers: Their population would increase because frogs, their predators, are gone, reducing predation pressure.
- Snakes: Their population would decrease because frogs are their primary food source. Without frogs, snakes would face food scarcity, leading to starvation or migration.
Why: The food chain shows frogs eat grasshoppers, and snakes eat frogs. Removing frogs disrupts the balance, allowing grasshoppers to proliferate and starving snakes (Page 10, Fig. 12.9).
Answer: Grasshopper population would increase due to lack of predation; snake population would decrease due to lack of food.
Q5: In a school garden, students noticed fewer butterflies the previous season. What could be the possible reasons? What steps can students take to have more butterflies on campus?
A5:
Possible reasons for fewer butterflies:
- Loss of host plants (e.g., milkweed) where butterflies lay eggs or feed.
- Pesticide use killing butterflies or their larvae.
- Reduced nectar sources due to fewer flowering plants.
- Habitat destruction from construction or landscaping.
- Increased predators (e.g., birds, dragonflies) or pollution affecting butterfly survival (Pages 6, 16).
Steps to increase butterflies: - Plant nectar-rich flowers (e.g., marigolds, zinnias) and host plants for larvae.
- Avoid synthetic pesticides; use natural pest control (e.g., ladybugs).
- Create a butterfly garden with diverse plants and water sources.
- Reduce mowing or clearing to preserve habitats.
- Educate the school community about butterfly conservation (Page 17).
Answer: Fewer butterflies could be due to loss of host plants, pesticides, or habitat destruction. Students can plant nectar and host plants, avoid pesticides, and create a butterfly garden.
Q6: Why is it not possible to have an ecosystem with only producers and no consumers or decomposers?
A6: An ecosystem with only producers (plants) is not possible because:
- Producers generate organic matter, but without consumers (herbivores, carnivores, omnivores), this matter would accumulate, leading to resource depletion (e.g., soil nutrients).
- Without decomposers, dead plant material would not break down, halting nutrient recycling, which plants need for growth.
- The absence of consumers and decomposers would disrupt energy flow and nutrient cycling, collapsing the ecosystem (Pages 9, 11).
Answer: Producers alone cannot sustain an ecosystem because consumers transfer energy, and decomposers recycle nutrients, both essential for ecosystem balance.
Q7: Observe two different places near your home or school (e.g., a park and a roadside). List the living and non-living components you see. How are the two ecosystems different?
A7:
Example observations:
- Park:
- Biotic: Trees, grass, birds, squirrels, insects, butterflies.
- Abiotic: Soil, water (pond or sprinklers), sunlight, air, benches.
- Roadside:
- Biotic: Weeds, stray dogs, ants, occasional birds.
- Abiotic: Soil, rocks, asphalt, air, sunlight, litter (plastic, paper).
Differences:
- Park: A managed ecosystem with diverse plants and animals, more water sources (e.g., ponds), and stable conditions supporting biodiversity.
- Roadside: A disturbed ecosystem with fewer species, dominated by hardy weeds and tolerant animals, affected by pollution, traffic, and less vegetation (Page 8).
Answer: Park has diverse biotic components and stable abiotic conditions; roadside has fewer species, more pollution, and disturbed conditions.
Q8: Human-made ecosystems like agricultural fields are necessary, but they must be made sustainable. Comment on the statement.
A8: Human-made ecosystems like agricultural fields are essential for food production to support growing populations. However, practices like monoculture, overuse of synthetic fertilizers, and pesticides degrade soil, reduce biodiversity, and harm ecosystems (e.g., killing pollinators, polluting water). Sustainable practices, such as organic farming, crop rotation, and natural pest control, maintain soil health, support biodiversity, and ensure long-term food security with minimal environmental impact (Pages 16-17).
Answer: Agricultural fields are necessary for food but must adopt sustainable practices like organic farming to protect soil, biodiversity, and ecosystems.
Q9: If the Indian hare population (Fig. 12.20) drops because of a disease, how would it affect the number of other organisms?
A9: If the Indian hare population drops:
- Herbivores (e.g., deer): May face increased competition for plants, as hares no longer consume them, potentially reducing plant diversity.
- Predators (e.g., foxes, eagles): Their populations would decrease due to food scarcity, as hares are a primary food source.
- Plants: May initially increase due to less grazing but could face overgrazing by other herbivores.
- Decomposers: May see less activity initially due to fewer hare carcasses but could stabilize as other populations adjust. The food web would be disrupted, affecting ecosystem balance (Page 11, Fig. 12.11).
Answer: Predators like foxes would decrease; plants may increase initially but face overgrazing; other herbivores may compete more, disrupting the food web.
Chapter 13 – Our Home: Earth, a Unique Life Sustaining Planet
Exercise Questions and Answers
Q1: What is one major reason Mars cannot currently support life like Earth?
(i) It has too many volcanoes.
(ii) It is too close to the Sun.
(iii) It lacks a thick atmosphere and liquid water.
(iv) Its magnetic field is too strong.
A1: Mars cannot support life like Earth because it lacks a thick atmosphere and liquid water, essential for life as we know it. Its thin atmosphere (100 times thinner than Earth’s) and position at the edge of the habitable zone limit liquid water availability (Pages 6-7).
Answer: (iii) It lacks a thick atmosphere and liquid water.
Q2: Which of these is an example of geodiversity?
(i) Variety of bird chirping in a forest.
(ii) Different landforms like mountains, valleys, and deserts.
(iii) Changing weather during monsoons.
(iv) Number of different types of fish in a pond.
A2: Geodiversity refers to the variety of landforms, rocks, and soils. Different landforms like mountains, valleys, and deserts are examples of geodiversity, as they create unique habitats (Page 10, Fig. 13.9).
Answer: (ii) Different landforms like mountains, valleys, and deserts.
Q3: If the Earth were smaller with the same density, what might happen to its atmosphere?
(i) It would become thicker and hotter.
(ii) It would escape into space due to weaker gravity.
(iii) It would become frozen.
(iv) It would cause stronger winds.
A3: A smaller Earth with the same density would have weaker gravity, unable to retain atmospheric gases, causing them to escape into space, as seen with Mars (Page 7).
Answer: (ii) It would escape into space due to weaker gravity.
Q4: In sexual reproduction, why are offspring different from their parents?
(i) They grow in different climates.
(ii) They eat different food.
(iii) They acquire new instructions after birth.
(iv) They get mixed instructions (genes) from both parents.
A4: Offspring in sexual reproduction differ from parents because they receive a mix of genetic instructions (genes) from both parents via gametes, creating unique combinations of traits (Page 13).
Answer: (iv) They get mixed instructions (genes) from both parents.
Q5: You notice tiny green plants growing on cracks on your school wall after the monsoon. Where do you think the seeds came from? What conditions helped these plants grow there?
A5:
- Seed origin: Seeds likely came from nearby plants, carried by wind, birds, or animals (e.g., birds eating fruits and excreting seeds). They settled in wall cracks (Page 13).
- Conditions for growth: Monsoon rains provided water, and cracks retained moisture and soil particles. Sunlight and nutrients in the cracks supported seed germination and growth, as learned in seed germination studies (Page 13).
Answer: Seeds were carried by wind, birds, or animals. Monsoon water, retained moisture, sunlight, and nutrients in cracks helped plants grow.
Q6: A city has recently cut down a large patch of forest to build new roads and buildings. Discuss the possible effects this could have on the local climate and biodiversity? How might this affect water availability or quality in the area?
A6:
- Local climate: Deforestation reduces trees that absorb carbon dioxide and produce oxygen, increasing local temperatures (less shade) and altering rainfall patterns due to reduced transpiration (Page 10).
- Biodiversity: Loss of forest habitat displaces or endangers animals, plants, and microbes, reducing species diversity and disrupting food webs (e.g., herbivores lose food, affecting predators) (Page 14).
- Water availability/quality: Fewer trees lead to soil erosion, reducing water retention and increasing runoff, which lowers groundwater levels. Runoff may carry pollutants from construction, degrading water quality in nearby rivers or lakes (Pages 10, 15).
Answer: Deforestation raises temperatures, alters rainfall, reduces biodiversity by displacing species, lowers groundwater due to erosion, and degrades water quality with pollutants.
Q7: A friend says, “The Earth has always had climate changes in the past, so today’s global warming is nothing new.” How would you respond using what you’ve learnt in this and other chapters of your science book?
A7: While Earth has experienced natural climate changes over millions of years (e.g., ice ages), today’s global warming is different because it is primarily driven by human activities, like burning fossil fuels, which release excessive greenhouse gases (carbon dioxide, methane). These gases intensify the greenhouse effect, causing rapid warming, melting ice caps, rising sea levels, and extreme weather, unlike slower natural changes. Deforestation and pollution further exacerbate this, threatening ecosystems and biodiversity (Pages 14-15, Chapter 12). The Paris Agreement (2015) highlights the urgency to limit warming to 1.5°C, but current efforts are insufficient, making human-induced climate change a unique and urgent crisis (Page 15).
Answer: Today’s global warming is human-driven, caused by fossil fuel emissions and deforestation, leading to rapid, severe changes unlike slow natural climate shifts, threatening ecosystems and requiring urgent action.
Q8. Imagine Earth’s magnetic field suddenly disappeared. What kinds of problems could arise for life on Earth? Explain.
A8: The Earth’s magnetic field acts like a protective shield. If it suddenly disappeared, these problems could arise:
1. Radiation from the Sun (Solar Wind)
- The magnetic field deflects charged particles from the Sun (solar wind).
- Without it, these harmful particles would directly strike Earth’s atmosphere.
- This could increase exposure to radiation, affecting humans, animals, and plants (higher risks of cancer, DNA damage).
2. Loss of Atmosphere
- Over long times, solar wind could strip away Earth’s atmosphere, just like it happened on Mars.
- This would reduce oxygen and make Earth less habitable.
3. Disruption of Technology
- Satellites and communication systems rely on protection from magnetic shielding.
- Without it, solar storms could cause power grid failures, GPS errors, and satellite damage.
4. Auroras Everywhere
- Normally, auroras (Northern and Southern Lights) are limited to polar regions where charged particles interact with the atmosphere.
- Without a magnetic field, auroras could appear across the globe – beautiful, but a sign of dangerous solar radiation.
5. Navigation Problems
- Many animals (like birds, turtles, and even bees) use Earth’s magnetic field to migrate.
- Without it, their navigation systems would fail, disturbing ecosystems.
Q9. You are tasked with designing a new settlement for humans on Mars. Name three things you would need to recreate from Earth to support human life there. Which of these do you think is the hardest to replicate, and why?
A9: Let’s imagine we are building a human settlement on Mars. To make it habitable, we’d need to recreate some key Earth-like conditions:
Three things needed:
- Breathable Atmosphere
- Mars has a very thin atmosphere, made mostly of carbon dioxide (CO₂).
- We need to create enough oxygen for humans to breathe, and maintain air pressure similar to Earth.
- Liquid Water Supply
- Water is essential for drinking, farming, hygiene, and producing oxygen + rocket fuel (via electrolysis).
- On Mars, water exists mostly as ice, so we’d need systems to extract, melt, and purify it.
- Suitable Temperature & Protection
- Mars is extremely cold (average −60 °C) and has no strong magnetic field to protect against harmful solar radiation.
- We’d need insulated habitats, greenhouses, and radiation shielding (underground bases or domes with protective layers).
Hardest to Replicate- The hardest to recreate is a protective atmosphere + magnetic shielding.
Reason:
- Oxygen can be produced (from CO₂ or water) with technology.
- Water can be extracted from Martian ice.
- But creating a thick, stable atmosphere with radiation protection requires altering the entire Martian environment (a huge task, called terraforming).
- Without this shield, humans must live in sealed domes or underground, relying on artificial systems.
Q10. In a village, the temperature has been increasing and rainfall has become unpredictable over the past few years. What could be causing this change? Suggest two ways the village could adapt to these new conditions.
A10: What could be causing the change?
- Global Climate Change: Rising greenhouse gases (like CO₂, methane) trap heat, leading to higher temperatures and disturbed rainfall patterns.
- Local Causes:
- Cutting down trees (deforestation) reduces rainfall and increases heat.
- Overuse of groundwater and poor farming practices dry up soil.
- Pollution from industries or vehicles can also affect local weather.
Two ways the village could adapt:
- Water Management & Conservation
- Build rainwater harvesting systems, ponds, or check dams to store rain when it does fall.
- Use drip irrigation and water-efficient crops to reduce wastage.
- Climate-Resilient Farming
- Shift to drought-resistant or short-duration crops that need less water.
- Practice crop diversification so that even if rains fail, some crops survive.
- Plant more trees (agroforestry) to cool the local environment and improve rainfall.
Q11. If there were no atmosphere on the Earth, would it affect life, temperature, and water on the planet? Explain.
A11: Yes , if Earth had no atmosphere, it would affect life, temperature, and water very severely.
Effect on Life
- The atmosphere provides oxygen for breathing and carbon dioxide for plants.
- Without it, no plants, animals, or humans could survive.
- Harmful solar radiation (UV rays, cosmic rays) would directly hit Earth, making it impossible for life to exist on the surface.
Effect on Temperature
- The atmosphere traps heat through the greenhouse effect, keeping Earth warm.
- Without it, daytime temperatures would become extremely hot (like the Moon, ~120 °C in sunlight) and nights extremely cold (−170 °C).
- This huge fluctuation would make Earth unlivable.
Effect on Water
- The atmosphere helps maintain water in liquid form by controlling pressure and temperature.
- Without it, liquid water would boil away or freeze, leaving Earth dry and barren.
Q12. Discuss five examples of vegetative propagation.
A12: Five Examples of Vegetative Propagation:
- Potato (Stem Tubers)
- New potato plants grow from the “eyes” (buds) on the tuber.
- Farmers use pieces of potato with eyes for planting.
- Onion & Garlic (Bulbs)
- Each bulb contains a short stem and fleshy leaves.
- When planted, the bulb grows into a new plant.
- Ginger & Turmeric (Rhizomes)
- These underground stems store food and have buds.
- Buds sprout into new shoots and roots, forming new plants.
- Bryophyllum (Leaf Buds)
- Small buds grow on the edges of leaves.
- When leaves fall on soil, the buds grow into new plants.
- Sugarcane (Stem Cuttings / Setts)
- Sugarcane stems have nodes with buds.
- Farmers cut the stem into pieces and plant them; each node can grow into a new plant.
🎉 Congratulations! We have successfully completed all the chapters of the NCERT Solutions for Class 8 Science Curiosity book. We hope this journey has not only helped you find the right answers but also deepened your understanding and curiosity for science.
💡 Remember — learning is not just about scoring well in exams, it’s about exploring, questioning, and growing every day.
If you have any queries or doubts, drop them in the comments below or write us an email — we’ll be happy to help!
📢 Don’t forget to share this content with your friends and classmates so they too can benefit and learn along with you. Together, we grow stronger!

